A Targeted Thorium-227 Conjugate Demonstrates Efficacy in Preclinical Models of Acquired Drug Resistance and Combination Potential with Chemotherapeutics and Antiangiogenic Therapies Article Swipe
 YOU?
·
· 2023
· Open Access
·
· DOI: https://doi.org/10.1158/1535-7163.mct-22-0808
YOU?
·
· 2023
· Open Access
·
· DOI: https://doi.org/10.1158/1535-7163.mct-22-0808
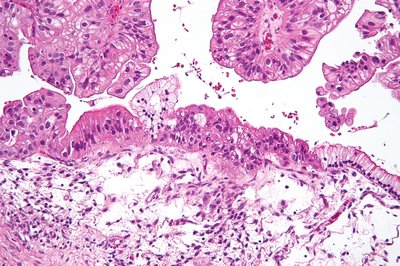
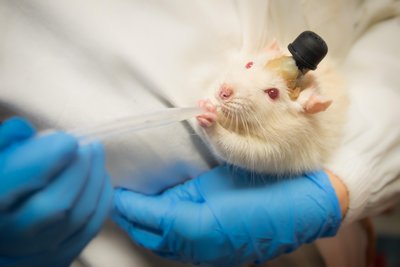
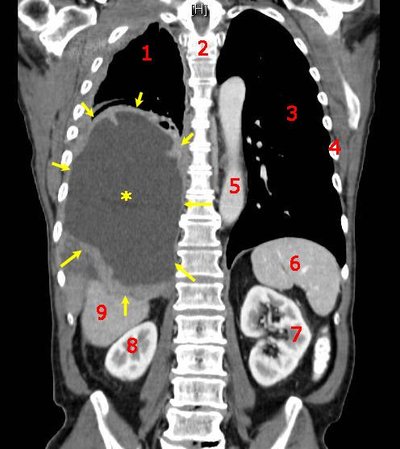
Targeted alpha therapies (TAT) are an innovative class of therapies for cancer treatment. The unique mode-of-action of TATs is the induction of deleterious DNA double-strand breaks. Difficult-to-treat cancers, such as gynecologic cancers upregulating the chemoresistance P-glycoprotein (p-gp) and overexpressing the membrane protein mesothelin (MSLN), are promising targets for TATs. Here, based on the previous encouraging findings with monotherapy, we investigated the efficacy of the mesothelin-targeted thorium-227 conjugate (MSLN-TTC) both as monotherapy and in combination with chemotherapies and antiangiogenic compounds in ovarian and cervical cancer models expressing p-gp. MSLN-TTC monotherapy showed equal cytotoxicity in vitro in p-gp–positive and -negative cancer cells, while chemotherapeutics dramatically lost activity on p-gp–positive cancer cells. In vivo, MSLN-TTC exhibited dose-dependent tumor growth inhibition with treatment/control ratios of 0.03–0.44 in various xenograft models irrespective of p-gp expression status. Furthermore, MSLN-TTC was more efficacious in p-gp–expressing tumors than chemotherapeutics. In the MSLN-expressing ST206B ovarian cancer patient-derived xenograft model, MSLN-TTC accumulated specifically in the tumor, which combined with pegylated liposomal doxorubicin (Doxil), docetaxel, bevacizumab, or regorafenib treatment induced additive-to-synergistic antitumor efficacy and substantially increased response rates compared with respective monotherapies. The combination treatments were well tolerated and only transient decreases in white and red blood cells were observed. In summary, we demonstrate that MSLN-TTC treatment shows efficacy in p-gp–expressing models of chemoresistance and has combination potential with chemo- and antiangiogenic therapies.
Related Topics
- Type
- article
- Language
- en
- Landing Page
- https://doi.org/10.1158/1535-7163.mct-22-0808
- https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdf
- OA Status
- hybrid
- Cited By
- 4
- References
- 54
- Related Works
- 10
- OpenAlex ID
- https://openalex.org/W4382044804
Raw OpenAlex JSON
- OpenAlex ID
-
https://openalex.org/W4382044804Canonical identifier for this work in OpenAlex
- DOI
-
https://doi.org/10.1158/1535-7163.mct-22-0808Digital Object Identifier
- Title
-
A Targeted Thorium-227 Conjugate Demonstrates Efficacy in Preclinical Models of Acquired Drug Resistance and Combination Potential with Chemotherapeutics and Antiangiogenic TherapiesWork title
- Type
-
articleOpenAlex work type
- Language
-
enPrimary language
- Publication year
-
2023Year of publication
- Publication date
-
2023-06-26Full publication date if available
- Authors
-
Sabine Zitzmann-Kolbe, Alexander Kristian, Dieter Zopf, Claudia Kamfenkel, Oliver Politz, Christine Ellingsen, Jochen Hilbig, Mark U. Juul, Jesper Fonslet, Carsten H. Nielsen, Christoph A. Schatz, Roger M. Bjerke, Alan Cuthbertson, Dominik Mumberg, Urs B. HagemannList of authors in order
- Landing page
-
https://doi.org/10.1158/1535-7163.mct-22-0808Publisher landing page
- PDF URL
-
https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdfDirect link to full text PDF
- Open access
-
YesWhether a free full text is available
- OA status
-
hybridOpen access status per OpenAlex
- OA URL
-
https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdfDirect OA link when available
- Concepts
-
Mesothelin, Medicine, Ovarian cancer, Cancer research, Bevacizumab, In vivo, Pharmacology, Olaparib, Carboplatin, Paclitaxel, Doxorubicin, Cytotoxicity, Cancer, In vitro, Chemotherapy, Internal medicine, Chemistry, Cisplatin, Biology, Biochemistry, Gene, Polymerase, Poly ADP ribose polymerase, BiotechnologyTop concepts (fields/topics) attached by OpenAlex
- Cited by
-
4Total citation count in OpenAlex
- Citations by year (recent)
-
2025: 3, 2024: 1Per-year citation counts (last 5 years)
- References (count)
-
54Number of works referenced by this work
- Related works (count)
-
10Other works algorithmically related by OpenAlex
Full payload
| id | https://openalex.org/W4382044804 |
|---|---|
| doi | https://doi.org/10.1158/1535-7163.mct-22-0808 |
| ids.doi | https://doi.org/10.1158/1535-7163.mct-22-0808 |
| ids.pmid | https://pubmed.ncbi.nlm.nih.gov/37365121 |
| ids.openalex | https://openalex.org/W4382044804 |
| fwci | 1.41221759 |
| mesh[0].qualifier_ui | |
| mesh[0].descriptor_ui | D006801 |
| mesh[0].is_major_topic | False |
| mesh[0].qualifier_name | |
| mesh[0].descriptor_name | Humans |
| mesh[1].qualifier_ui | |
| mesh[1].descriptor_ui | D005260 |
| mesh[1].is_major_topic | False |
| mesh[1].qualifier_name | |
| mesh[1].descriptor_name | Female |
| mesh[2].qualifier_ui | |
| mesh[2].descriptor_ui | D000090204 |
| mesh[2].is_major_topic | True |
| mesh[2].qualifier_name | |
| mesh[2].descriptor_name | Mesothelin |
| mesh[3].qualifier_ui | |
| mesh[3].descriptor_ui | D058851 |
| mesh[3].is_major_topic | False |
| mesh[3].qualifier_name | |
| mesh[3].descriptor_name | GPI-Linked Proteins |
| mesh[4].qualifier_ui | |
| mesh[4].descriptor_ui | D045744 |
| mesh[4].is_major_topic | False |
| mesh[4].qualifier_name | |
| mesh[4].descriptor_name | Cell Line, Tumor |
| mesh[5].qualifier_ui | |
| mesh[5].descriptor_ui | D004351 |
| mesh[5].is_major_topic | False |
| mesh[5].qualifier_name | |
| mesh[5].descriptor_name | Drug Resistance |
| mesh[6].qualifier_ui | |
| mesh[6].descriptor_ui | D006801 |
| mesh[6].is_major_topic | False |
| mesh[6].qualifier_name | |
| mesh[6].descriptor_name | Humans |
| mesh[7].qualifier_ui | |
| mesh[7].descriptor_ui | D005260 |
| mesh[7].is_major_topic | False |
| mesh[7].qualifier_name | |
| mesh[7].descriptor_name | Female |
| mesh[8].qualifier_ui | |
| mesh[8].descriptor_ui | D000090204 |
| mesh[8].is_major_topic | True |
| mesh[8].qualifier_name | |
| mesh[8].descriptor_name | Mesothelin |
| mesh[9].qualifier_ui | |
| mesh[9].descriptor_ui | D058851 |
| mesh[9].is_major_topic | False |
| mesh[9].qualifier_name | |
| mesh[9].descriptor_name | GPI-Linked Proteins |
| mesh[10].qualifier_ui | |
| mesh[10].descriptor_ui | D045744 |
| mesh[10].is_major_topic | False |
| mesh[10].qualifier_name | |
| mesh[10].descriptor_name | Cell Line, Tumor |
| mesh[11].qualifier_ui | |
| mesh[11].descriptor_ui | D004351 |
| mesh[11].is_major_topic | False |
| mesh[11].qualifier_name | |
| mesh[11].descriptor_name | Drug Resistance |
| type | article |
| title | A Targeted Thorium-227 Conjugate Demonstrates Efficacy in Preclinical Models of Acquired Drug Resistance and Combination Potential with Chemotherapeutics and Antiangiogenic Therapies |
| biblio.issue | 9 |
| biblio.volume | 22 |
| biblio.last_page | 1086 |
| biblio.first_page | 1073 |
| topics[0].id | https://openalex.org/T10754 |
| topics[0].field.id | https://openalex.org/fields/27 |
| topics[0].field.display_name | Medicine |
| topics[0].score | 0.9977999925613403 |
| topics[0].domain.id | https://openalex.org/domains/4 |
| topics[0].domain.display_name | Health Sciences |
| topics[0].subfield.id | https://openalex.org/subfields/2713 |
| topics[0].subfield.display_name | Epidemiology |
| topics[0].display_name | Neuroendocrine Tumor Research Advances |
| topics[1].id | https://openalex.org/T10231 |
| topics[1].field.id | https://openalex.org/fields/27 |
| topics[1].field.display_name | Medicine |
| topics[1].score | 0.9958999752998352 |
| topics[1].domain.id | https://openalex.org/domains/4 |
| topics[1].domain.display_name | Health Sciences |
| topics[1].subfield.id | https://openalex.org/subfields/2730 |
| topics[1].subfield.display_name | Oncology |
| topics[1].display_name | Pancreatic and Hepatic Oncology Research |
| topics[2].id | https://openalex.org/T12334 |
| topics[2].field.id | https://openalex.org/fields/27 |
| topics[2].field.display_name | Medicine |
| topics[2].score | 0.9955000281333923 |
| topics[2].domain.id | https://openalex.org/domains/4 |
| topics[2].domain.display_name | Health Sciences |
| topics[2].subfield.id | https://openalex.org/subfields/2730 |
| topics[2].subfield.display_name | Oncology |
| topics[2].display_name | Lung Cancer Research Studies |
| is_xpac | False |
| apc_list | |
| apc_paid | |
| concepts[0].id | https://openalex.org/C2777045440 |
| concepts[0].level | 3 |
| concepts[0].score | 0.8959981203079224 |
| concepts[0].wikidata | https://www.wikidata.org/wiki/Q14914057 |
| concepts[0].display_name | Mesothelin |
| concepts[1].id | https://openalex.org/C71924100 |
| concepts[1].level | 0 |
| concepts[1].score | 0.6375223994255066 |
| concepts[1].wikidata | https://www.wikidata.org/wiki/Q11190 |
| concepts[1].display_name | Medicine |
| concepts[2].id | https://openalex.org/C2780427987 |
| concepts[2].level | 3 |
| concepts[2].score | 0.6107223629951477 |
| concepts[2].wikidata | https://www.wikidata.org/wiki/Q172341 |
| concepts[2].display_name | Ovarian cancer |
| concepts[3].id | https://openalex.org/C502942594 |
| concepts[3].level | 1 |
| concepts[3].score | 0.6073113679885864 |
| concepts[3].wikidata | https://www.wikidata.org/wiki/Q3421914 |
| concepts[3].display_name | Cancer research |
| concepts[4].id | https://openalex.org/C2777802072 |
| concepts[4].level | 3 |
| concepts[4].score | 0.5828565359115601 |
| concepts[4].wikidata | https://www.wikidata.org/wiki/Q413299 |
| concepts[4].display_name | Bevacizumab |
| concepts[5].id | https://openalex.org/C207001950 |
| concepts[5].level | 2 |
| concepts[5].score | 0.5013480186462402 |
| concepts[5].wikidata | https://www.wikidata.org/wiki/Q141124 |
| concepts[5].display_name | In vivo |
| concepts[6].id | https://openalex.org/C98274493 |
| concepts[6].level | 1 |
| concepts[6].score | 0.49404457211494446 |
| concepts[6].wikidata | https://www.wikidata.org/wiki/Q128406 |
| concepts[6].display_name | Pharmacology |
| concepts[7].id | https://openalex.org/C2779962180 |
| concepts[7].level | 5 |
| concepts[7].score | 0.4795805513858795 |
| concepts[7].wikidata | https://www.wikidata.org/wiki/Q7083106 |
| concepts[7].display_name | Olaparib |
| concepts[8].id | https://openalex.org/C2781451048 |
| concepts[8].level | 4 |
| concepts[8].score | 0.4442172050476074 |
| concepts[8].wikidata | https://www.wikidata.org/wiki/Q415588 |
| concepts[8].display_name | Carboplatin |
| concepts[9].id | https://openalex.org/C2777292972 |
| concepts[9].level | 3 |
| concepts[9].score | 0.4440500736236572 |
| concepts[9].wikidata | https://www.wikidata.org/wiki/Q423762 |
| concepts[9].display_name | Paclitaxel |
| concepts[10].id | https://openalex.org/C2781303535 |
| concepts[10].level | 3 |
| concepts[10].score | 0.44307181239128113 |
| concepts[10].wikidata | https://www.wikidata.org/wiki/Q18936 |
| concepts[10].display_name | Doxorubicin |
| concepts[11].id | https://openalex.org/C109316439 |
| concepts[11].level | 3 |
| concepts[11].score | 0.43220895528793335 |
| concepts[11].wikidata | https://www.wikidata.org/wiki/Q246181 |
| concepts[11].display_name | Cytotoxicity |
| concepts[12].id | https://openalex.org/C121608353 |
| concepts[12].level | 2 |
| concepts[12].score | 0.4095809757709503 |
| concepts[12].wikidata | https://www.wikidata.org/wiki/Q12078 |
| concepts[12].display_name | Cancer |
| concepts[13].id | https://openalex.org/C202751555 |
| concepts[13].level | 2 |
| concepts[13].score | 0.3491475582122803 |
| concepts[13].wikidata | https://www.wikidata.org/wiki/Q221681 |
| concepts[13].display_name | In vitro |
| concepts[14].id | https://openalex.org/C2776694085 |
| concepts[14].level | 2 |
| concepts[14].score | 0.2837908864021301 |
| concepts[14].wikidata | https://www.wikidata.org/wiki/Q974135 |
| concepts[14].display_name | Chemotherapy |
| concepts[15].id | https://openalex.org/C126322002 |
| concepts[15].level | 1 |
| concepts[15].score | 0.22218286991119385 |
| concepts[15].wikidata | https://www.wikidata.org/wiki/Q11180 |
| concepts[15].display_name | Internal medicine |
| concepts[16].id | https://openalex.org/C185592680 |
| concepts[16].level | 0 |
| concepts[16].score | 0.1830410659313202 |
| concepts[16].wikidata | https://www.wikidata.org/wiki/Q2329 |
| concepts[16].display_name | Chemistry |
| concepts[17].id | https://openalex.org/C2778239845 |
| concepts[17].level | 3 |
| concepts[17].score | 0.1515476107597351 |
| concepts[17].wikidata | https://www.wikidata.org/wiki/Q412415 |
| concepts[17].display_name | Cisplatin |
| concepts[18].id | https://openalex.org/C86803240 |
| concepts[18].level | 0 |
| concepts[18].score | 0.135964035987854 |
| concepts[18].wikidata | https://www.wikidata.org/wiki/Q420 |
| concepts[18].display_name | Biology |
| concepts[19].id | https://openalex.org/C55493867 |
| concepts[19].level | 1 |
| concepts[19].score | 0.0 |
| concepts[19].wikidata | https://www.wikidata.org/wiki/Q7094 |
| concepts[19].display_name | Biochemistry |
| concepts[20].id | https://openalex.org/C104317684 |
| concepts[20].level | 2 |
| concepts[20].score | 0.0 |
| concepts[20].wikidata | https://www.wikidata.org/wiki/Q7187 |
| concepts[20].display_name | Gene |
| concepts[21].id | https://openalex.org/C82381507 |
| concepts[21].level | 3 |
| concepts[21].score | 0.0 |
| concepts[21].wikidata | https://www.wikidata.org/wiki/Q416878 |
| concepts[21].display_name | Polymerase |
| concepts[22].id | https://openalex.org/C182979987 |
| concepts[22].level | 4 |
| concepts[22].score | 0.0 |
| concepts[22].wikidata | https://www.wikidata.org/wiki/Q3907241 |
| concepts[22].display_name | Poly ADP ribose polymerase |
| concepts[23].id | https://openalex.org/C150903083 |
| concepts[23].level | 1 |
| concepts[23].score | 0.0 |
| concepts[23].wikidata | https://www.wikidata.org/wiki/Q7108 |
| concepts[23].display_name | Biotechnology |
| keywords[0].id | https://openalex.org/keywords/mesothelin |
| keywords[0].score | 0.8959981203079224 |
| keywords[0].display_name | Mesothelin |
| keywords[1].id | https://openalex.org/keywords/medicine |
| keywords[1].score | 0.6375223994255066 |
| keywords[1].display_name | Medicine |
| keywords[2].id | https://openalex.org/keywords/ovarian-cancer |
| keywords[2].score | 0.6107223629951477 |
| keywords[2].display_name | Ovarian cancer |
| keywords[3].id | https://openalex.org/keywords/cancer-research |
| keywords[3].score | 0.6073113679885864 |
| keywords[3].display_name | Cancer research |
| keywords[4].id | https://openalex.org/keywords/bevacizumab |
| keywords[4].score | 0.5828565359115601 |
| keywords[4].display_name | Bevacizumab |
| keywords[5].id | https://openalex.org/keywords/in-vivo |
| keywords[5].score | 0.5013480186462402 |
| keywords[5].display_name | In vivo |
| keywords[6].id | https://openalex.org/keywords/pharmacology |
| keywords[6].score | 0.49404457211494446 |
| keywords[6].display_name | Pharmacology |
| keywords[7].id | https://openalex.org/keywords/olaparib |
| keywords[7].score | 0.4795805513858795 |
| keywords[7].display_name | Olaparib |
| keywords[8].id | https://openalex.org/keywords/carboplatin |
| keywords[8].score | 0.4442172050476074 |
| keywords[8].display_name | Carboplatin |
| keywords[9].id | https://openalex.org/keywords/paclitaxel |
| keywords[9].score | 0.4440500736236572 |
| keywords[9].display_name | Paclitaxel |
| keywords[10].id | https://openalex.org/keywords/doxorubicin |
| keywords[10].score | 0.44307181239128113 |
| keywords[10].display_name | Doxorubicin |
| keywords[11].id | https://openalex.org/keywords/cytotoxicity |
| keywords[11].score | 0.43220895528793335 |
| keywords[11].display_name | Cytotoxicity |
| keywords[12].id | https://openalex.org/keywords/cancer |
| keywords[12].score | 0.4095809757709503 |
| keywords[12].display_name | Cancer |
| keywords[13].id | https://openalex.org/keywords/in-vitro |
| keywords[13].score | 0.3491475582122803 |
| keywords[13].display_name | In vitro |
| keywords[14].id | https://openalex.org/keywords/chemotherapy |
| keywords[14].score | 0.2837908864021301 |
| keywords[14].display_name | Chemotherapy |
| keywords[15].id | https://openalex.org/keywords/internal-medicine |
| keywords[15].score | 0.22218286991119385 |
| keywords[15].display_name | Internal medicine |
| keywords[16].id | https://openalex.org/keywords/chemistry |
| keywords[16].score | 0.1830410659313202 |
| keywords[16].display_name | Chemistry |
| keywords[17].id | https://openalex.org/keywords/cisplatin |
| keywords[17].score | 0.1515476107597351 |
| keywords[17].display_name | Cisplatin |
| keywords[18].id | https://openalex.org/keywords/biology |
| keywords[18].score | 0.135964035987854 |
| keywords[18].display_name | Biology |
| language | en |
| locations[0].id | doi:10.1158/1535-7163.mct-22-0808 |
| locations[0].is_oa | True |
| locations[0].source.id | https://openalex.org/S48437117 |
| locations[0].source.issn | 1535-7163, 1538-8514 |
| locations[0].source.type | journal |
| locations[0].source.is_oa | False |
| locations[0].source.issn_l | 1535-7163 |
| locations[0].source.is_core | True |
| locations[0].source.is_in_doaj | False |
| locations[0].source.display_name | Molecular Cancer Therapeutics |
| locations[0].source.host_organization | https://openalex.org/P4310320273 |
| locations[0].source.host_organization_name | American Association for Cancer Research |
| locations[0].source.host_organization_lineage | https://openalex.org/P4310320273 |
| locations[0].license | cc-by-nc-nd |
| locations[0].pdf_url | https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdf |
| locations[0].version | publishedVersion |
| locations[0].raw_type | journal-article |
| locations[0].license_id | https://openalex.org/licenses/cc-by-nc-nd |
| locations[0].is_accepted | True |
| locations[0].is_published | True |
| locations[0].raw_source_name | Molecular Cancer Therapeutics |
| locations[0].landing_page_url | https://doi.org/10.1158/1535-7163.mct-22-0808 |
| locations[1].id | pmid:37365121 |
| locations[1].is_oa | False |
| locations[1].source.id | https://openalex.org/S4306525036 |
| locations[1].source.issn | |
| locations[1].source.type | repository |
| locations[1].source.is_oa | False |
| locations[1].source.issn_l | |
| locations[1].source.is_core | False |
| locations[1].source.is_in_doaj | False |
| locations[1].source.display_name | PubMed |
| locations[1].source.host_organization | https://openalex.org/I1299303238 |
| locations[1].source.host_organization_name | National Institutes of Health |
| locations[1].source.host_organization_lineage | https://openalex.org/I1299303238 |
| locations[1].license | |
| locations[1].pdf_url | |
| locations[1].version | publishedVersion |
| locations[1].raw_type | |
| locations[1].license_id | |
| locations[1].is_accepted | True |
| locations[1].is_published | True |
| locations[1].raw_source_name | Molecular cancer therapeutics |
| locations[1].landing_page_url | https://pubmed.ncbi.nlm.nih.gov/37365121 |
| locations[2].id | pmh:oai:pubmedcentral.nih.gov:10477831 |
| locations[2].is_oa | True |
| locations[2].source.id | https://openalex.org/S2764455111 |
| locations[2].source.issn | |
| locations[2].source.type | repository |
| locations[2].source.is_oa | False |
| locations[2].source.issn_l | |
| locations[2].source.is_core | False |
| locations[2].source.is_in_doaj | False |
| locations[2].source.display_name | PubMed Central |
| locations[2].source.host_organization | https://openalex.org/I1299303238 |
| locations[2].source.host_organization_name | National Institutes of Health |
| locations[2].source.host_organization_lineage | https://openalex.org/I1299303238 |
| locations[2].license | cc-by-nc-nd |
| locations[2].pdf_url | https://pmc.ncbi.nlm.nih.gov/articles/PMC10477831/pdf/1073.pdf |
| locations[2].version | submittedVersion |
| locations[2].raw_type | Text |
| locations[2].license_id | https://openalex.org/licenses/cc-by-nc-nd |
| locations[2].is_accepted | False |
| locations[2].is_published | False |
| locations[2].raw_source_name | Mol Cancer Ther |
| locations[2].landing_page_url | https://www.ncbi.nlm.nih.gov/pmc/articles/10477831 |
| indexed_in | crossref, pubmed |
| authorships[0].author.id | https://openalex.org/A5082071316 |
| authorships[0].author.orcid | https://orcid.org/0000-0002-1476-6299 |
| authorships[0].author.display_name | Sabine Zitzmann-Kolbe |
| authorships[0].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[0].author_position | first |
| authorships[0].raw_author_name | Sabine Zitzmann-Kolbe |
| authorships[0].is_corresponding | False |
| authorships[0].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[1].author.id | https://openalex.org/A5058059489 |
| authorships[1].author.orcid | |
| authorships[1].author.display_name | Alexander Kristian |
| authorships[1].affiliations[0].raw_affiliation_string | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[1].author_position | middle |
| authorships[1].raw_author_name | Alexander Kristian |
| authorships[1].is_corresponding | False |
| authorships[1].raw_affiliation_strings | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[2].author.id | https://openalex.org/A5034373024 |
| authorships[2].author.orcid | https://orcid.org/0000-0001-9251-7792 |
| authorships[2].author.display_name | Dieter Zopf |
| authorships[2].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[2].author_position | middle |
| authorships[2].raw_author_name | Dieter Zopf |
| authorships[2].is_corresponding | False |
| authorships[2].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[3].author.id | https://openalex.org/A5005148708 |
| authorships[3].author.orcid | https://orcid.org/0009-0005-9578-7751 |
| authorships[3].author.display_name | Claudia Kamfenkel |
| authorships[3].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[3].author_position | middle |
| authorships[3].raw_author_name | Claudia Kamfenkel |
| authorships[3].is_corresponding | False |
| authorships[3].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[4].author.id | https://openalex.org/A5056198089 |
| authorships[4].author.orcid | https://orcid.org/0000-0002-0272-6249 |
| authorships[4].author.display_name | Oliver Politz |
| authorships[4].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[4].author_position | middle |
| authorships[4].raw_author_name | Oliver Politz |
| authorships[4].is_corresponding | False |
| authorships[4].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[5].author.id | https://openalex.org/A5006335909 |
| authorships[5].author.orcid | https://orcid.org/0009-0006-4567-9354 |
| authorships[5].author.display_name | Christine Ellingsen |
| authorships[5].affiliations[0].raw_affiliation_string | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[5].author_position | middle |
| authorships[5].raw_author_name | Christine Ellingsen |
| authorships[5].is_corresponding | False |
| authorships[5].raw_affiliation_strings | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[6].author.id | https://openalex.org/A5016955536 |
| authorships[6].author.orcid | https://orcid.org/0009-0005-0379-9465 |
| authorships[6].author.display_name | Jochen Hilbig |
| authorships[6].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[6].author_position | middle |
| authorships[6].raw_author_name | Jochen Hilbig |
| authorships[6].is_corresponding | False |
| authorships[6].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[7].author.id | https://openalex.org/A5033560120 |
| authorships[7].author.orcid | https://orcid.org/0009-0008-3194-9709 |
| authorships[7].author.display_name | Mark U. Juul |
| authorships[7].affiliations[0].raw_affiliation_string | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[7].author_position | middle |
| authorships[7].raw_author_name | Mark U. Juul |
| authorships[7].is_corresponding | False |
| authorships[7].raw_affiliation_strings | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[8].author.id | https://openalex.org/A5072570184 |
| authorships[8].author.orcid | https://orcid.org/0000-0003-1563-0251 |
| authorships[8].author.display_name | Jesper Fonslet |
| authorships[8].affiliations[0].raw_affiliation_string | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[8].author_position | middle |
| authorships[8].raw_author_name | Jesper Fonslet |
| authorships[8].is_corresponding | False |
| authorships[8].raw_affiliation_strings | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[9].author.id | https://openalex.org/A5006783007 |
| authorships[9].author.orcid | https://orcid.org/0009-0009-6632-7835 |
| authorships[9].author.display_name | Carsten H. Nielsen |
| authorships[9].affiliations[0].raw_affiliation_string | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[9].author_position | middle |
| authorships[9].raw_author_name | Carsten H. Nielsen |
| authorships[9].is_corresponding | False |
| authorships[9].raw_affiliation_strings | 3Minerva Imaging ApS, Oelstykke, Denmark. |
| authorships[10].author.id | https://openalex.org/A5017327688 |
| authorships[10].author.orcid | https://orcid.org/0000-0001-8028-2631 |
| authorships[10].author.display_name | Christoph A. Schatz |
| authorships[10].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[10].author_position | middle |
| authorships[10].raw_author_name | Christoph A. Schatz |
| authorships[10].is_corresponding | False |
| authorships[10].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[11].author.id | https://openalex.org/A5037599842 |
| authorships[11].author.orcid | https://orcid.org/0009-0006-0678-5517 |
| authorships[11].author.display_name | Roger M. Bjerke |
| authorships[11].affiliations[0].raw_affiliation_string | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[11].author_position | middle |
| authorships[11].raw_author_name | Roger M. Bjerke |
| authorships[11].is_corresponding | False |
| authorships[11].raw_affiliation_strings | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[12].author.id | https://openalex.org/A5063128206 |
| authorships[12].author.orcid | https://orcid.org/0009-0000-4887-9761 |
| authorships[12].author.display_name | Alan Cuthbertson |
| authorships[12].affiliations[0].raw_affiliation_string | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[12].author_position | middle |
| authorships[12].raw_author_name | Alan S. Cuthbertson |
| authorships[12].is_corresponding | False |
| authorships[12].raw_affiliation_strings | 2Bayer AS, Thorium Conjugate Research, Oslo, Norway. |
| authorships[13].author.id | https://openalex.org/A5009359386 |
| authorships[13].author.orcid | https://orcid.org/0000-0002-6670-1497 |
| authorships[13].author.display_name | Dominik Mumberg |
| authorships[13].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[13].author_position | middle |
| authorships[13].raw_author_name | Dominik Mumberg |
| authorships[13].is_corresponding | False |
| authorships[13].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[14].author.id | https://openalex.org/A5072394114 |
| authorships[14].author.orcid | https://orcid.org/0009-0003-6509-0844 |
| authorships[14].author.display_name | Urs B. Hagemann |
| authorships[14].affiliations[0].raw_affiliation_string | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| authorships[14].author_position | last |
| authorships[14].raw_author_name | Urs B. Hagemann |
| authorships[14].is_corresponding | False |
| authorships[14].raw_affiliation_strings | 1Bayer AG, Pharmaceuticals, Berlin, Germany. |
| has_content.pdf | True |
| has_content.grobid_xml | True |
| is_paratext | False |
| open_access.is_oa | True |
| open_access.oa_url | https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdf |
| open_access.oa_status | hybrid |
| open_access.any_repository_has_fulltext | False |
| created_date | 2025-10-10T00:00:00 |
| display_name | A Targeted Thorium-227 Conjugate Demonstrates Efficacy in Preclinical Models of Acquired Drug Resistance and Combination Potential with Chemotherapeutics and Antiangiogenic Therapies |
| has_fulltext | True |
| is_retracted | False |
| updated_date | 2025-11-23T05:10:03.516525 |
| primary_topic.id | https://openalex.org/T10754 |
| primary_topic.field.id | https://openalex.org/fields/27 |
| primary_topic.field.display_name | Medicine |
| primary_topic.score | 0.9977999925613403 |
| primary_topic.domain.id | https://openalex.org/domains/4 |
| primary_topic.domain.display_name | Health Sciences |
| primary_topic.subfield.id | https://openalex.org/subfields/2713 |
| primary_topic.subfield.display_name | Epidemiology |
| primary_topic.display_name | Neuroendocrine Tumor Research Advances |
| related_works | https://openalex.org/W2093727844, https://openalex.org/W4214953906, https://openalex.org/W4213459853, https://openalex.org/W4214780373, https://openalex.org/W2416964709, https://openalex.org/W1885581526, https://openalex.org/W4213392866, https://openalex.org/W4240540248, https://openalex.org/W2015548499, https://openalex.org/W248568554 |
| cited_by_count | 4 |
| counts_by_year[0].year | 2025 |
| counts_by_year[0].cited_by_count | 3 |
| counts_by_year[1].year | 2024 |
| counts_by_year[1].cited_by_count | 1 |
| locations_count | 3 |
| best_oa_location.id | doi:10.1158/1535-7163.mct-22-0808 |
| best_oa_location.is_oa | True |
| best_oa_location.source.id | https://openalex.org/S48437117 |
| best_oa_location.source.issn | 1535-7163, 1538-8514 |
| best_oa_location.source.type | journal |
| best_oa_location.source.is_oa | False |
| best_oa_location.source.issn_l | 1535-7163 |
| best_oa_location.source.is_core | True |
| best_oa_location.source.is_in_doaj | False |
| best_oa_location.source.display_name | Molecular Cancer Therapeutics |
| best_oa_location.source.host_organization | https://openalex.org/P4310320273 |
| best_oa_location.source.host_organization_name | American Association for Cancer Research |
| best_oa_location.source.host_organization_lineage | https://openalex.org/P4310320273 |
| best_oa_location.license | cc-by-nc-nd |
| best_oa_location.pdf_url | https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdf |
| best_oa_location.version | publishedVersion |
| best_oa_location.raw_type | journal-article |
| best_oa_location.license_id | https://openalex.org/licenses/cc-by-nc-nd |
| best_oa_location.is_accepted | True |
| best_oa_location.is_published | True |
| best_oa_location.raw_source_name | Molecular Cancer Therapeutics |
| best_oa_location.landing_page_url | https://doi.org/10.1158/1535-7163.mct-22-0808 |
| primary_location.id | doi:10.1158/1535-7163.mct-22-0808 |
| primary_location.is_oa | True |
| primary_location.source.id | https://openalex.org/S48437117 |
| primary_location.source.issn | 1535-7163, 1538-8514 |
| primary_location.source.type | journal |
| primary_location.source.is_oa | False |
| primary_location.source.issn_l | 1535-7163 |
| primary_location.source.is_core | True |
| primary_location.source.is_in_doaj | False |
| primary_location.source.display_name | Molecular Cancer Therapeutics |
| primary_location.source.host_organization | https://openalex.org/P4310320273 |
| primary_location.source.host_organization_name | American Association for Cancer Research |
| primary_location.source.host_organization_lineage | https://openalex.org/P4310320273 |
| primary_location.license | cc-by-nc-nd |
| primary_location.pdf_url | https://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0808/3343751/mct-22-0808.pdf |
| primary_location.version | publishedVersion |
| primary_location.raw_type | journal-article |
| primary_location.license_id | https://openalex.org/licenses/cc-by-nc-nd |
| primary_location.is_accepted | True |
| primary_location.is_published | True |
| primary_location.raw_source_name | Molecular Cancer Therapeutics |
| primary_location.landing_page_url | https://doi.org/10.1158/1535-7163.mct-22-0808 |
| publication_date | 2023-06-26 |
| publication_year | 2023 |
| referenced_works | https://openalex.org/W2891647981, https://openalex.org/W6775600713, https://openalex.org/W2944344108, https://openalex.org/W2921520187, https://openalex.org/W2254640582, https://openalex.org/W2996077865, https://openalex.org/W3164081628, https://openalex.org/W3128646645, https://openalex.org/W3092122627, https://openalex.org/W2965404853, https://openalex.org/W4211203170, https://openalex.org/W2917476978, https://openalex.org/W6683134993, https://openalex.org/W2794128499, https://openalex.org/W2753722921, https://openalex.org/W2772038399, https://openalex.org/W4220785729, https://openalex.org/W3082918087, https://openalex.org/W3149678565, https://openalex.org/W2992107509, https://openalex.org/W1968209756, https://openalex.org/W2560499218, https://openalex.org/W2116922255, https://openalex.org/W2147834058, https://openalex.org/W4310051805, https://openalex.org/W2897984531, https://openalex.org/W2138654358, https://openalex.org/W2103562758, https://openalex.org/W2060993743, https://openalex.org/W2019607817, https://openalex.org/W2022095438, https://openalex.org/W2772085397, https://openalex.org/W2094586578, https://openalex.org/W2111358509, https://openalex.org/W2489035057, https://openalex.org/W2088659772, https://openalex.org/W7074107537, https://openalex.org/W2038320131, https://openalex.org/W2035248522, https://openalex.org/W2016318120, https://openalex.org/W2154200023, https://openalex.org/W1981376295, https://openalex.org/W2050912869, https://openalex.org/W2044774111, https://openalex.org/W6641105450, https://openalex.org/W2168709731, https://openalex.org/W2161962458, https://openalex.org/W2792755823, https://openalex.org/W6692199315, https://openalex.org/W3203731043, https://openalex.org/W1954597686, https://openalex.org/W1972160506, https://openalex.org/W3015002607, https://openalex.org/W2591655368 |
| referenced_works_count | 54 |
| abstract_inverted_index.In | 110, 142, 200 |
| abstract_inverted_index.an | 6 |
| abstract_inverted_index.as | 30, 70 |
| abstract_inverted_index.in | 73, 80, 93, 95, 123, 137, 154, 192, 209 |
| abstract_inverted_index.is | 19 |
| abstract_inverted_index.of | 9, 17, 22, 63, 121, 128, 212 |
| abstract_inverted_index.on | 52, 106 |
| abstract_inverted_index.or | 166 |
| abstract_inverted_index.we | 59, 202 |
| abstract_inverted_index.DNA | 24 |
| abstract_inverted_index.The | 14, 182 |
| abstract_inverted_index.and | 38, 72, 77, 82, 97, 173, 188, 194, 214, 220 |
| abstract_inverted_index.are | 5, 45 |
| abstract_inverted_index.for | 11, 48 |
| abstract_inverted_index.has | 215 |
| abstract_inverted_index.red | 195 |
| abstract_inverted_index.the | 20, 34, 40, 53, 61, 64, 143, 155 |
| abstract_inverted_index.was | 134 |
| abstract_inverted_index.TATs | 18 |
| abstract_inverted_index.both | 69 |
| abstract_inverted_index.lost | 104 |
| abstract_inverted_index.more | 135 |
| abstract_inverted_index.only | 189 |
| abstract_inverted_index.p-gp | 129 |
| abstract_inverted_index.such | 29 |
| abstract_inverted_index.than | 140 |
| abstract_inverted_index.that | 204 |
| abstract_inverted_index.well | 186 |
| abstract_inverted_index.were | 185, 198 |
| abstract_inverted_index.with | 57, 75, 118, 159, 179, 218 |
| abstract_inverted_index.(TAT) | 4 |
| abstract_inverted_index.Here, | 50 |
| abstract_inverted_index.TATs. | 49 |
| abstract_inverted_index.alpha | 2 |
| abstract_inverted_index.based | 51 |
| abstract_inverted_index.blood | 196 |
| abstract_inverted_index.cells | 197 |
| abstract_inverted_index.class | 8 |
| abstract_inverted_index.equal | 91 |
| abstract_inverted_index.p-gp. | 87 |
| abstract_inverted_index.rates | 177 |
| abstract_inverted_index.shows | 207 |
| abstract_inverted_index.tumor | 115 |
| abstract_inverted_index.vitro | 94 |
| abstract_inverted_index.vivo, | 111 |
| abstract_inverted_index.which | 157 |
| abstract_inverted_index.while | 101 |
| abstract_inverted_index.white | 193 |
| abstract_inverted_index.(p-gp) | 37 |
| abstract_inverted_index.ST206B | 145 |
| abstract_inverted_index.cancer | 12, 84, 99, 108, 147 |
| abstract_inverted_index.cells, | 100 |
| abstract_inverted_index.cells. | 109 |
| abstract_inverted_index.chemo- | 219 |
| abstract_inverted_index.growth | 116 |
| abstract_inverted_index.model, | 150 |
| abstract_inverted_index.models | 85, 126, 211 |
| abstract_inverted_index.ratios | 120 |
| abstract_inverted_index.showed | 90 |
| abstract_inverted_index.tumor, | 156 |
| abstract_inverted_index.tumors | 139 |
| abstract_inverted_index.unique | 15 |
| abstract_inverted_index.(MSLN), | 44 |
| abstract_inverted_index.breaks. | 26 |
| abstract_inverted_index.cancers | 32 |
| abstract_inverted_index.induced | 169 |
| abstract_inverted_index.ovarian | 81, 146 |
| abstract_inverted_index.protein | 42 |
| abstract_inverted_index.status. | 131 |
| abstract_inverted_index.targets | 47 |
| abstract_inverted_index.various | 124 |
| abstract_inverted_index.(Doxil), | 163 |
| abstract_inverted_index.Abstract | 0 |
| abstract_inverted_index.MSLN-TTC | 88, 112, 133, 151, 205 |
| abstract_inverted_index.Targeted | 1 |
| abstract_inverted_index.activity | 105 |
| abstract_inverted_index.cancers, | 28 |
| abstract_inverted_index.cervical | 83 |
| abstract_inverted_index.combined | 158 |
| abstract_inverted_index.compared | 178 |
| abstract_inverted_index.efficacy | 62, 172, 208 |
| abstract_inverted_index.findings | 56 |
| abstract_inverted_index.membrane | 41 |
| abstract_inverted_index.previous | 54 |
| abstract_inverted_index.response | 176 |
| abstract_inverted_index.summary, | 201 |
| abstract_inverted_index.-negative | 98 |
| abstract_inverted_index.antitumor | 171 |
| abstract_inverted_index.compounds | 79 |
| abstract_inverted_index.conjugate | 67 |
| abstract_inverted_index.decreases | 191 |
| abstract_inverted_index.exhibited | 113 |
| abstract_inverted_index.increased | 175 |
| abstract_inverted_index.induction | 21 |
| abstract_inverted_index.liposomal | 161 |
| abstract_inverted_index.observed. | 199 |
| abstract_inverted_index.pegylated | 160 |
| abstract_inverted_index.potential | 217 |
| abstract_inverted_index.promising | 46 |
| abstract_inverted_index.therapies | 3, 10 |
| abstract_inverted_index.tolerated | 187 |
| abstract_inverted_index.transient | 190 |
| abstract_inverted_index.treatment | 168, 206 |
| abstract_inverted_index.xenograft | 125, 149 |
| abstract_inverted_index.(MSLN-TTC) | 68 |
| abstract_inverted_index.docetaxel, | 164 |
| abstract_inverted_index.expressing | 86 |
| abstract_inverted_index.expression | 130 |
| abstract_inverted_index.inhibition | 117 |
| abstract_inverted_index.innovative | 7 |
| abstract_inverted_index.mesothelin | 43 |
| abstract_inverted_index.respective | 180 |
| abstract_inverted_index.therapies. | 222 |
| abstract_inverted_index.treatment. | 13 |
| abstract_inverted_index.treatments | 184 |
| abstract_inverted_index.0.03–0.44 | 122 |
| abstract_inverted_index.accumulated | 152 |
| abstract_inverted_index.combination | 74, 183, 216 |
| abstract_inverted_index.deleterious | 23 |
| abstract_inverted_index.demonstrate | 203 |
| abstract_inverted_index.doxorubicin | 162 |
| abstract_inverted_index.efficacious | 136 |
| abstract_inverted_index.encouraging | 55 |
| abstract_inverted_index.gynecologic | 31 |
| abstract_inverted_index.monotherapy | 71, 89 |
| abstract_inverted_index.regorafenib | 167 |
| abstract_inverted_index.thorium-227 | 66 |
| abstract_inverted_index.Furthermore, | 132 |
| abstract_inverted_index.bevacizumab, | 165 |
| abstract_inverted_index.cytotoxicity | 92 |
| abstract_inverted_index.dramatically | 103 |
| abstract_inverted_index.investigated | 60 |
| abstract_inverted_index.irrespective | 127 |
| abstract_inverted_index.monotherapy, | 58 |
| abstract_inverted_index.specifically | 153 |
| abstract_inverted_index.upregulating | 33 |
| abstract_inverted_index.double-strand | 25 |
| abstract_inverted_index.substantially | 174 |
| abstract_inverted_index.P-glycoprotein | 36 |
| abstract_inverted_index.antiangiogenic | 78, 221 |
| abstract_inverted_index.chemotherapies | 76 |
| abstract_inverted_index.dose-dependent | 114 |
| abstract_inverted_index.mode-of-action | 16 |
| abstract_inverted_index.monotherapies. | 181 |
| abstract_inverted_index.overexpressing | 39 |
| abstract_inverted_index.MSLN-expressing | 144 |
| abstract_inverted_index.chemoresistance | 35, 213 |
| abstract_inverted_index.p-gp–positive | 96, 107 |
| abstract_inverted_index.patient-derived | 148 |
| abstract_inverted_index.chemotherapeutics | 102 |
| abstract_inverted_index.p-gp–expressing | 138, 210 |
| abstract_inverted_index.treatment/control | 119 |
| abstract_inverted_index.Difficult-to-treat | 27 |
| abstract_inverted_index.chemotherapeutics. | 141 |
| abstract_inverted_index.mesothelin-targeted | 65 |
| abstract_inverted_index.additive-to-synergistic | 170 |
| cited_by_percentile_year.max | 98 |
| cited_by_percentile_year.min | 90 |
| countries_distinct_count | 0 |
| institutions_distinct_count | 15 |
| sustainable_development_goals[0].id | https://metadata.un.org/sdg/3 |
| sustainable_development_goals[0].score | 0.8399999737739563 |
| sustainable_development_goals[0].display_name | Good health and well-being |
| citation_normalized_percentile.value | 0.77778148 |
| citation_normalized_percentile.is_in_top_1_percent | False |
| citation_normalized_percentile.is_in_top_10_percent | False |