Berberine Ursodeoxycholate for the Treatment of Type 2 Diabetes Article Swipe
 YOU?
·
· 2025
· Open Access
·
· DOI: https://doi.org/10.1001/jamanetworkopen.2024.62185
YOU?
·
· 2025
· Open Access
·
· DOI: https://doi.org/10.1001/jamanetworkopen.2024.62185
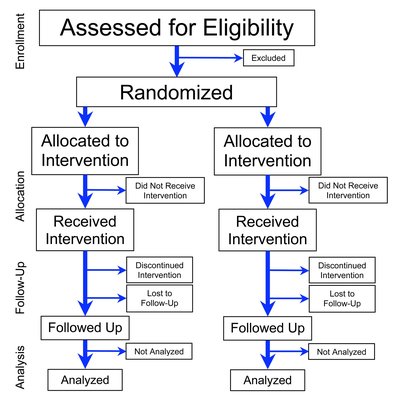
Importance Few of the available therapies for type 2 diabetes (T2D) comprehensively address disease burden beyond glycemic control. Examining whether berberine ursodeoxycholate (HTD1801), a first-in-class gut-liver anti-inflammatory metabolic modulator, has the potential to treat the core aspects of metabolic disease is important. Objective To assess the safety and efficacy of HTD1801 in patients with T2D that is inadequately controlled with diet and exercise. Design, Setting, and Participants This phase 2 double-blind, placebo-controlled, 12-week randomized clinical trial, conducted in China between March 2022 and January 2023, included patients with T2D who underwent 8 or more weeks of diet and exercise, had a hemoglobin A 1c (HbA 1c ) level of 7.0% to 10.5%, and had a fasting plasma glucose (FPG) level less than 250.5 mg/dL. Interventions Patients were randomized 1:1:1 to placebo (n = 38), HTD1801 500 mg twice daily (n = 37), and HTD1801 1000 mg twice daily (n = 38). Main Outcomes and Measures The primary end point was the HbA 1c level change from baseline to week 12. Secondary end points included glycemic, hepatic, and cardiometabolic parameters. The primary end point was analyzed using a mixed-effects model for repeated measures, with the HbA 1c level change from baseline as the dependent variable. Treatment group, measurement time point, and interaction between treatment group and measurement time point were independent variables. Results The study included 113 patients with T2D (mean [SD] age, 54.3 [10.6] years; 72 male [63.7%]) who were randomized. Among these patients, the mean (SD) HbA 1c level was 8.2% (0.8%); body mass index, 25.5 (3.7), calculated as weight in kilograms divided by height in meters squared; and FPG level, 160.7 (38.3) mg/dL. Baseline disease severity was balanced across treatment groups. The primary end point was achieved with significant dose-dependent reductions in the HbA 1c level in both HTD1801 groups compared with the placebo group. The least-squares mean difference in the HbA 1c level at week 12 was −0.4% (95% CI, −0.79% to −0.03%; P = .04) for the 500-mg group and −0.7% (95% CI, −1.10% to −0.35%; P < .001) for the 1000-mg group compared with the placebo group. HbA 1c level reductions were paralleled with mean (SD) improvements in the FPG level in both the 500-mg group (−13.0 [38.2] mg/dL) and the 1000-mg group (−18.4 [21.8] mg/dL) groups. Reductions were observed in lipids and markers of liver injury in the 1000-mg group. HTD1801 was safe and well tolerated, with 110 patients (97.3%) completing the study. Treatment-emergent adverse events, generally mild, occurred in 59 patients (52.2%) overall. One patient (in the 500-mg group) experienced a serious adverse event of retinal hemorrhage, which was unlikely related to treatment. No patients discontinued due to an adverse event. Conclusions and Relevance In this placebo-controlled randomized clinical trial, treatment with HTD1801 resulted in significant reductions in the HbA 1c level and improvements in key cardiometabolic and liver parameters. HTD1801 was safe and well tolerated. These findings are being confirmed in ongoing phase 3 studies. The effects demonstrated by HTD1801 support an oral treatment option for T2D and its comorbidities. Trial Registration ClinicalTrials.gov Identifier: NCT06411275
Related Topics
- Type
- article
- Language
- en
- Landing Page
- https://doi.org/10.1001/jamanetworkopen.2024.62185
- https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdf
- OA Status
- gold
- Cited By
- 11
- References
- 36
- Related Works
- 10
- OpenAlex ID
- https://openalex.org/W4408091434
Raw OpenAlex JSON
- OpenAlex ID
-
https://openalex.org/W4408091434Canonical identifier for this work in OpenAlex
- DOI
-
https://doi.org/10.1001/jamanetworkopen.2024.62185Digital Object Identifier
- Title
-
Berberine Ursodeoxycholate for the Treatment of Type 2 DiabetesWork title
- Type
-
articleOpenAlex work type
- Language
-
enPrimary language
- Publication year
-
2025Year of publication
- Publication date
-
2025-03-03Full publication date if available
- Authors
-
Linong Ji, Jianhua Ma, Yujin Ma, Zhifeng Cheng, Shenglian Gan, Guoyue Yuan, Dexue Liu, Sheli Li, Yu Liu, Xia Xue, Jie Bai, Kun Wang, Hanqing Cai, Shu Li, Kui Liu, Meng Yu, Liping LiuList of authors in order
- Landing page
-
https://doi.org/10.1001/jamanetworkopen.2024.62185Publisher landing page
- PDF URL
-
https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdfDirect link to full text PDF
- Open access
-
YesWhether a free full text is available
- OA status
-
goldOpen access status per OpenAlex
- OA URL
-
https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdfDirect OA link when available
- Concepts
-
Medicine, Glycemic, Type 2 diabetes, Internal medicine, Clinical endpoint, Randomized controlled trial, Placebo, Diabetes mellitus, Physical therapy, Endocrinology, Insulin, Alternative medicine, PathologyTop concepts (fields/topics) attached by OpenAlex
- Cited by
-
11Total citation count in OpenAlex
- Citations by year (recent)
-
2025: 11Per-year citation counts (last 5 years)
- References (count)
-
36Number of works referenced by this work
- Related works (count)
-
10Other works algorithmically related by OpenAlex
Full payload
| id | https://openalex.org/W4408091434 |
|---|---|
| doi | https://doi.org/10.1001/jamanetworkopen.2024.62185 |
| ids.doi | https://doi.org/10.1001/jamanetworkopen.2024.62185 |
| ids.pmid | https://pubmed.ncbi.nlm.nih.gov/40029660 |
| ids.openalex | https://openalex.org/W4408091434 |
| fwci | 53.07318082 |
| mesh[0].qualifier_ui | |
| mesh[0].descriptor_ui | D006801 |
| mesh[0].is_major_topic | False |
| mesh[0].qualifier_name | |
| mesh[0].descriptor_name | Humans |
| mesh[1].qualifier_ui | Q000188 |
| mesh[1].descriptor_ui | D003924 |
| mesh[1].is_major_topic | True |
| mesh[1].qualifier_name | drug therapy |
| mesh[1].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[2].qualifier_ui | Q000097 |
| mesh[2].descriptor_ui | D003924 |
| mesh[2].is_major_topic | True |
| mesh[2].qualifier_name | blood |
| mesh[2].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[3].qualifier_ui | |
| mesh[3].descriptor_ui | D008297 |
| mesh[3].is_major_topic | False |
| mesh[3].qualifier_name | |
| mesh[3].descriptor_name | Male |
| mesh[4].qualifier_ui | |
| mesh[4].descriptor_ui | D008875 |
| mesh[4].is_major_topic | False |
| mesh[4].qualifier_name | |
| mesh[4].descriptor_name | Middle Aged |
| mesh[5].qualifier_ui | |
| mesh[5].descriptor_ui | D005260 |
| mesh[5].is_major_topic | False |
| mesh[5].qualifier_name | |
| mesh[5].descriptor_name | Female |
| mesh[6].qualifier_ui | |
| mesh[6].descriptor_ui | D004311 |
| mesh[6].is_major_topic | False |
| mesh[6].qualifier_name | |
| mesh[6].descriptor_name | Double-Blind Method |
| mesh[7].qualifier_ui | Q000627 |
| mesh[7].descriptor_ui | D001599 |
| mesh[7].is_major_topic | True |
| mesh[7].qualifier_name | therapeutic use |
| mesh[7].descriptor_name | Berberine |
| mesh[8].qualifier_ui | Q000008 |
| mesh[8].descriptor_ui | D001599 |
| mesh[8].is_major_topic | True |
| mesh[8].qualifier_name | administration & dosage |
| mesh[8].descriptor_name | Berberine |
| mesh[9].qualifier_ui | Q000032 |
| mesh[9].descriptor_ui | D006442 |
| mesh[9].is_major_topic | False |
| mesh[9].qualifier_name | analysis |
| mesh[9].descriptor_name | Glycated Hemoglobin |
| mesh[10].qualifier_ui | Q000187 |
| mesh[10].descriptor_ui | D006442 |
| mesh[10].is_major_topic | False |
| mesh[10].qualifier_name | drug effects |
| mesh[10].descriptor_name | Glycated Hemoglobin |
| mesh[11].qualifier_ui | Q000187 |
| mesh[11].descriptor_ui | D001786 |
| mesh[11].is_major_topic | False |
| mesh[11].qualifier_name | drug effects |
| mesh[11].descriptor_name | Blood Glucose |
| mesh[12].qualifier_ui | Q000032 |
| mesh[12].descriptor_ui | D001786 |
| mesh[12].is_major_topic | False |
| mesh[12].qualifier_name | analysis |
| mesh[12].descriptor_name | Blood Glucose |
| mesh[13].qualifier_ui | |
| mesh[13].descriptor_ui | D000328 |
| mesh[13].is_major_topic | False |
| mesh[13].qualifier_name | |
| mesh[13].descriptor_name | Adult |
| mesh[14].qualifier_ui | |
| mesh[14].descriptor_ui | D000368 |
| mesh[14].is_major_topic | False |
| mesh[14].qualifier_name | |
| mesh[14].descriptor_name | Aged |
| mesh[15].qualifier_ui | |
| mesh[15].descriptor_ui | D016896 |
| mesh[15].is_major_topic | False |
| mesh[15].qualifier_name | |
| mesh[15].descriptor_name | Treatment Outcome |
| mesh[16].qualifier_ui | Q000627 |
| mesh[16].descriptor_ui | D007004 |
| mesh[16].is_major_topic | False |
| mesh[16].qualifier_name | therapeutic use |
| mesh[16].descriptor_name | Hypoglycemic Agents |
| mesh[17].qualifier_ui | |
| mesh[17].descriptor_ui | D002681 |
| mesh[17].is_major_topic | False |
| mesh[17].qualifier_name | |
| mesh[17].descriptor_name | China |
| mesh[18].qualifier_ui | |
| mesh[18].descriptor_ui | D006801 |
| mesh[18].is_major_topic | False |
| mesh[18].qualifier_name | |
| mesh[18].descriptor_name | Humans |
| mesh[19].qualifier_ui | Q000188 |
| mesh[19].descriptor_ui | D003924 |
| mesh[19].is_major_topic | True |
| mesh[19].qualifier_name | drug therapy |
| mesh[19].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[20].qualifier_ui | Q000097 |
| mesh[20].descriptor_ui | D003924 |
| mesh[20].is_major_topic | True |
| mesh[20].qualifier_name | blood |
| mesh[20].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[21].qualifier_ui | |
| mesh[21].descriptor_ui | D008297 |
| mesh[21].is_major_topic | False |
| mesh[21].qualifier_name | |
| mesh[21].descriptor_name | Male |
| mesh[22].qualifier_ui | |
| mesh[22].descriptor_ui | D008875 |
| mesh[22].is_major_topic | False |
| mesh[22].qualifier_name | |
| mesh[22].descriptor_name | Middle Aged |
| mesh[23].qualifier_ui | |
| mesh[23].descriptor_ui | D005260 |
| mesh[23].is_major_topic | False |
| mesh[23].qualifier_name | |
| mesh[23].descriptor_name | Female |
| mesh[24].qualifier_ui | |
| mesh[24].descriptor_ui | D004311 |
| mesh[24].is_major_topic | False |
| mesh[24].qualifier_name | |
| mesh[24].descriptor_name | Double-Blind Method |
| mesh[25].qualifier_ui | Q000627 |
| mesh[25].descriptor_ui | D001599 |
| mesh[25].is_major_topic | True |
| mesh[25].qualifier_name | therapeutic use |
| mesh[25].descriptor_name | Berberine |
| mesh[26].qualifier_ui | Q000008 |
| mesh[26].descriptor_ui | D001599 |
| mesh[26].is_major_topic | True |
| mesh[26].qualifier_name | administration & dosage |
| mesh[26].descriptor_name | Berberine |
| mesh[27].qualifier_ui | Q000032 |
| mesh[27].descriptor_ui | D006442 |
| mesh[27].is_major_topic | False |
| mesh[27].qualifier_name | analysis |
| mesh[27].descriptor_name | Glycated Hemoglobin |
| mesh[28].qualifier_ui | Q000187 |
| mesh[28].descriptor_ui | D006442 |
| mesh[28].is_major_topic | False |
| mesh[28].qualifier_name | drug effects |
| mesh[28].descriptor_name | Glycated Hemoglobin |
| mesh[29].qualifier_ui | Q000187 |
| mesh[29].descriptor_ui | D001786 |
| mesh[29].is_major_topic | False |
| mesh[29].qualifier_name | drug effects |
| mesh[29].descriptor_name | Blood Glucose |
| mesh[30].qualifier_ui | Q000032 |
| mesh[30].descriptor_ui | D001786 |
| mesh[30].is_major_topic | False |
| mesh[30].qualifier_name | analysis |
| mesh[30].descriptor_name | Blood Glucose |
| mesh[31].qualifier_ui | |
| mesh[31].descriptor_ui | D000328 |
| mesh[31].is_major_topic | False |
| mesh[31].qualifier_name | |
| mesh[31].descriptor_name | Adult |
| mesh[32].qualifier_ui | |
| mesh[32].descriptor_ui | D000368 |
| mesh[32].is_major_topic | False |
| mesh[32].qualifier_name | |
| mesh[32].descriptor_name | Aged |
| mesh[33].qualifier_ui | |
| mesh[33].descriptor_ui | D016896 |
| mesh[33].is_major_topic | False |
| mesh[33].qualifier_name | |
| mesh[33].descriptor_name | Treatment Outcome |
| mesh[34].qualifier_ui | Q000627 |
| mesh[34].descriptor_ui | D007004 |
| mesh[34].is_major_topic | False |
| mesh[34].qualifier_name | therapeutic use |
| mesh[34].descriptor_name | Hypoglycemic Agents |
| mesh[35].qualifier_ui | |
| mesh[35].descriptor_ui | D002681 |
| mesh[35].is_major_topic | False |
| mesh[35].qualifier_name | |
| mesh[35].descriptor_name | China |
| mesh[36].qualifier_ui | |
| mesh[36].descriptor_ui | D006801 |
| mesh[36].is_major_topic | False |
| mesh[36].qualifier_name | |
| mesh[36].descriptor_name | Humans |
| mesh[37].qualifier_ui | Q000188 |
| mesh[37].descriptor_ui | D003924 |
| mesh[37].is_major_topic | True |
| mesh[37].qualifier_name | drug therapy |
| mesh[37].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[38].qualifier_ui | Q000097 |
| mesh[38].descriptor_ui | D003924 |
| mesh[38].is_major_topic | True |
| mesh[38].qualifier_name | blood |
| mesh[38].descriptor_name | Diabetes Mellitus, Type 2 |
| mesh[39].qualifier_ui | |
| mesh[39].descriptor_ui | D008297 |
| mesh[39].is_major_topic | False |
| mesh[39].qualifier_name | |
| mesh[39].descriptor_name | Male |
| mesh[40].qualifier_ui | |
| mesh[40].descriptor_ui | D005260 |
| mesh[40].is_major_topic | False |
| mesh[40].qualifier_name | |
| mesh[40].descriptor_name | Female |
| mesh[41].qualifier_ui | |
| mesh[41].descriptor_ui | D008875 |
| mesh[41].is_major_topic | False |
| mesh[41].qualifier_name | |
| mesh[41].descriptor_name | Middle Aged |
| mesh[42].qualifier_ui | Q000627 |
| mesh[42].descriptor_ui | D001599 |
| mesh[42].is_major_topic | True |
| mesh[42].qualifier_name | therapeutic use |
| mesh[42].descriptor_name | Berberine |
| mesh[43].qualifier_ui | |
| mesh[43].descriptor_ui | D004311 |
| mesh[43].is_major_topic | False |
| mesh[43].qualifier_name | |
| mesh[43].descriptor_name | Double-Blind Method |
| mesh[44].qualifier_ui | Q000032 |
| mesh[44].descriptor_ui | D006442 |
| mesh[44].is_major_topic | False |
| mesh[44].qualifier_name | analysis |
| mesh[44].descriptor_name | Glycated Hemoglobin |
| mesh[45].qualifier_ui | Q000627 |
| mesh[45].descriptor_ui | D014580 |
| mesh[45].is_major_topic | True |
| mesh[45].qualifier_name | therapeutic use |
| mesh[45].descriptor_name | Ursodeoxycholic Acid |
| mesh[46].qualifier_ui | Q000008 |
| mesh[46].descriptor_ui | D014580 |
| mesh[46].is_major_topic | True |
| mesh[46].qualifier_name | administration & dosage |
| mesh[46].descriptor_name | Ursodeoxycholic Acid |
| mesh[47].qualifier_ui | |
| mesh[47].descriptor_ui | D000368 |
| mesh[47].is_major_topic | False |
| mesh[47].qualifier_name | |
| mesh[47].descriptor_name | Aged |
| mesh[48].qualifier_ui | Q000187 |
| mesh[48].descriptor_ui | D001786 |
| mesh[48].is_major_topic | False |
| mesh[48].qualifier_name | drug effects |
| mesh[48].descriptor_name | Blood Glucose |
| mesh[49].qualifier_ui | Q000032 |
| mesh[49].descriptor_ui | D001786 |
| mesh[49].is_major_topic | False |
| mesh[49].qualifier_name | analysis |
| mesh[49].descriptor_name | Blood Glucose |
| type | article |
| title | Berberine Ursodeoxycholate for the Treatment of Type 2 Diabetes |
| biblio.issue | 3 |
| biblio.volume | 8 |
| biblio.last_page | e2462185 |
| biblio.first_page | e2462185 |
| topics[0].id | https://openalex.org/T12059 |
| topics[0].field.id | https://openalex.org/fields/27 |
| topics[0].field.display_name | Medicine |
| topics[0].score | 0.9997000098228455 |
| topics[0].domain.id | https://openalex.org/domains/4 |
| topics[0].domain.display_name | Health Sciences |
| topics[0].subfield.id | https://openalex.org/subfields/2736 |
| topics[0].subfield.display_name | Pharmacology |
| topics[0].display_name | Berberine and alkaloids research |
| topics[1].id | https://openalex.org/T11140 |
| topics[1].field.id | https://openalex.org/fields/27 |
| topics[1].field.display_name | Medicine |
| topics[1].score | 0.9812999963760376 |
| topics[1].domain.id | https://openalex.org/domains/4 |
| topics[1].domain.display_name | Health Sciences |
| topics[1].subfield.id | https://openalex.org/subfields/2712 |
| topics[1].subfield.display_name | Endocrinology, Diabetes and Metabolism |
| topics[1].display_name | Natural Antidiabetic Agents Studies |
| topics[2].id | https://openalex.org/T10414 |
| topics[2].field.id | https://openalex.org/fields/27 |
| topics[2].field.display_name | Medicine |
| topics[2].score | 0.9797999858856201 |
| topics[2].domain.id | https://openalex.org/domains/4 |
| topics[2].domain.display_name | Health Sciences |
| topics[2].subfield.id | https://openalex.org/subfields/2736 |
| topics[2].subfield.display_name | Pharmacology |
| topics[2].display_name | Cannabis and Cannabinoid Research |
| is_xpac | False |
| apc_list.value | 3000 |
| apc_list.currency | USD |
| apc_list.value_usd | 3000 |
| apc_paid.value | 3000 |
| apc_paid.currency | USD |
| apc_paid.value_usd | 3000 |
| concepts[0].id | https://openalex.org/C71924100 |
| concepts[0].level | 0 |
| concepts[0].score | 0.8427178263664246 |
| concepts[0].wikidata | https://www.wikidata.org/wiki/Q11190 |
| concepts[0].display_name | Medicine |
| concepts[1].id | https://openalex.org/C2780473172 |
| concepts[1].level | 3 |
| concepts[1].score | 0.8090914487838745 |
| concepts[1].wikidata | https://www.wikidata.org/wiki/Q5572518 |
| concepts[1].display_name | Glycemic |
| concepts[2].id | https://openalex.org/C2777180221 |
| concepts[2].level | 3 |
| concepts[2].score | 0.7548913359642029 |
| concepts[2].wikidata | https://www.wikidata.org/wiki/Q3025883 |
| concepts[2].display_name | Type 2 diabetes |
| concepts[3].id | https://openalex.org/C126322002 |
| concepts[3].level | 1 |
| concepts[3].score | 0.6588609218597412 |
| concepts[3].wikidata | https://www.wikidata.org/wiki/Q11180 |
| concepts[3].display_name | Internal medicine |
| concepts[4].id | https://openalex.org/C203092338 |
| concepts[4].level | 3 |
| concepts[4].score | 0.6317008137702942 |
| concepts[4].wikidata | https://www.wikidata.org/wiki/Q1340863 |
| concepts[4].display_name | Clinical endpoint |
| concepts[5].id | https://openalex.org/C168563851 |
| concepts[5].level | 2 |
| concepts[5].score | 0.6181561946868896 |
| concepts[5].wikidata | https://www.wikidata.org/wiki/Q1436668 |
| concepts[5].display_name | Randomized controlled trial |
| concepts[6].id | https://openalex.org/C27081682 |
| concepts[6].level | 3 |
| concepts[6].score | 0.5883404016494751 |
| concepts[6].wikidata | https://www.wikidata.org/wiki/Q269829 |
| concepts[6].display_name | Placebo |
| concepts[7].id | https://openalex.org/C555293320 |
| concepts[7].level | 2 |
| concepts[7].score | 0.5406621694564819 |
| concepts[7].wikidata | https://www.wikidata.org/wiki/Q12206 |
| concepts[7].display_name | Diabetes mellitus |
| concepts[8].id | https://openalex.org/C1862650 |
| concepts[8].level | 1 |
| concepts[8].score | 0.3718469738960266 |
| concepts[8].wikidata | https://www.wikidata.org/wiki/Q186005 |
| concepts[8].display_name | Physical therapy |
| concepts[9].id | https://openalex.org/C134018914 |
| concepts[9].level | 1 |
| concepts[9].score | 0.28164827823638916 |
| concepts[9].wikidata | https://www.wikidata.org/wiki/Q162606 |
| concepts[9].display_name | Endocrinology |
| concepts[10].id | https://openalex.org/C2779306644 |
| concepts[10].level | 2 |
| concepts[10].score | 0.20609739422798157 |
| concepts[10].wikidata | https://www.wikidata.org/wiki/Q2002370 |
| concepts[10].display_name | Insulin |
| concepts[11].id | https://openalex.org/C204787440 |
| concepts[11].level | 2 |
| concepts[11].score | 0.0 |
| concepts[11].wikidata | https://www.wikidata.org/wiki/Q188504 |
| concepts[11].display_name | Alternative medicine |
| concepts[12].id | https://openalex.org/C142724271 |
| concepts[12].level | 1 |
| concepts[12].score | 0.0 |
| concepts[12].wikidata | https://www.wikidata.org/wiki/Q7208 |
| concepts[12].display_name | Pathology |
| keywords[0].id | https://openalex.org/keywords/medicine |
| keywords[0].score | 0.8427178263664246 |
| keywords[0].display_name | Medicine |
| keywords[1].id | https://openalex.org/keywords/glycemic |
| keywords[1].score | 0.8090914487838745 |
| keywords[1].display_name | Glycemic |
| keywords[2].id | https://openalex.org/keywords/type-2-diabetes |
| keywords[2].score | 0.7548913359642029 |
| keywords[2].display_name | Type 2 diabetes |
| keywords[3].id | https://openalex.org/keywords/internal-medicine |
| keywords[3].score | 0.6588609218597412 |
| keywords[3].display_name | Internal medicine |
| keywords[4].id | https://openalex.org/keywords/clinical-endpoint |
| keywords[4].score | 0.6317008137702942 |
| keywords[4].display_name | Clinical endpoint |
| keywords[5].id | https://openalex.org/keywords/randomized-controlled-trial |
| keywords[5].score | 0.6181561946868896 |
| keywords[5].display_name | Randomized controlled trial |
| keywords[6].id | https://openalex.org/keywords/placebo |
| keywords[6].score | 0.5883404016494751 |
| keywords[6].display_name | Placebo |
| keywords[7].id | https://openalex.org/keywords/diabetes-mellitus |
| keywords[7].score | 0.5406621694564819 |
| keywords[7].display_name | Diabetes mellitus |
| keywords[8].id | https://openalex.org/keywords/physical-therapy |
| keywords[8].score | 0.3718469738960266 |
| keywords[8].display_name | Physical therapy |
| keywords[9].id | https://openalex.org/keywords/endocrinology |
| keywords[9].score | 0.28164827823638916 |
| keywords[9].display_name | Endocrinology |
| keywords[10].id | https://openalex.org/keywords/insulin |
| keywords[10].score | 0.20609739422798157 |
| keywords[10].display_name | Insulin |
| language | en |
| locations[0].id | doi:10.1001/jamanetworkopen.2024.62185 |
| locations[0].is_oa | True |
| locations[0].source.id | https://openalex.org/S4210217848 |
| locations[0].source.issn | 2574-3805 |
| locations[0].source.type | journal |
| locations[0].source.is_oa | True |
| locations[0].source.issn_l | 2574-3805 |
| locations[0].source.is_core | True |
| locations[0].source.is_in_doaj | True |
| locations[0].source.display_name | JAMA Network Open |
| locations[0].source.host_organization | https://openalex.org/P4310320259 |
| locations[0].source.host_organization_name | American Medical Association |
| locations[0].source.host_organization_lineage | https://openalex.org/P4310320259 |
| locations[0].license | |
| locations[0].pdf_url | https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdf |
| locations[0].version | publishedVersion |
| locations[0].raw_type | journal-article |
| locations[0].license_id | |
| locations[0].is_accepted | True |
| locations[0].is_published | True |
| locations[0].raw_source_name | JAMA Network Open |
| locations[0].landing_page_url | https://doi.org/10.1001/jamanetworkopen.2024.62185 |
| locations[1].id | pmid:40029660 |
| locations[1].is_oa | False |
| locations[1].source.id | https://openalex.org/S4306525036 |
| locations[1].source.issn | |
| locations[1].source.type | repository |
| locations[1].source.is_oa | False |
| locations[1].source.issn_l | |
| locations[1].source.is_core | False |
| locations[1].source.is_in_doaj | False |
| locations[1].source.display_name | PubMed |
| locations[1].source.host_organization | https://openalex.org/I1299303238 |
| locations[1].source.host_organization_name | National Institutes of Health |
| locations[1].source.host_organization_lineage | https://openalex.org/I1299303238 |
| locations[1].license | |
| locations[1].pdf_url | |
| locations[1].version | publishedVersion |
| locations[1].raw_type | |
| locations[1].license_id | |
| locations[1].is_accepted | True |
| locations[1].is_published | True |
| locations[1].raw_source_name | JAMA network open |
| locations[1].landing_page_url | https://pubmed.ncbi.nlm.nih.gov/40029660 |
| locations[2].id | pmh:oai:pubmedcentral.nih.gov:11877176 |
| locations[2].is_oa | True |
| locations[2].source.id | https://openalex.org/S2764455111 |
| locations[2].source.issn | |
| locations[2].source.type | repository |
| locations[2].source.is_oa | False |
| locations[2].source.issn_l | |
| locations[2].source.is_core | False |
| locations[2].source.is_in_doaj | False |
| locations[2].source.display_name | PubMed Central |
| locations[2].source.host_organization | https://openalex.org/I1299303238 |
| locations[2].source.host_organization_name | National Institutes of Health |
| locations[2].source.host_organization_lineage | https://openalex.org/I1299303238 |
| locations[2].license | other-oa |
| locations[2].pdf_url | |
| locations[2].version | submittedVersion |
| locations[2].raw_type | Text |
| locations[2].license_id | https://openalex.org/licenses/other-oa |
| locations[2].is_accepted | False |
| locations[2].is_published | False |
| locations[2].raw_source_name | JAMA Netw Open |
| locations[2].landing_page_url | https://www.ncbi.nlm.nih.gov/pmc/articles/11877176 |
| indexed_in | crossref, doaj, pubmed |
| authorships[0].author.id | https://openalex.org/A5036949656 |
| authorships[0].author.orcid | https://orcid.org/0000-0003-1305-1598 |
| authorships[0].author.display_name | Linong Ji |
| authorships[0].countries | CN |
| authorships[0].affiliations[0].institution_ids | https://openalex.org/I20231570, https://openalex.org/I4210124809 |
| authorships[0].affiliations[0].raw_affiliation_string | Department of Endocrinology, Peking University People's Hospital, Beijing, China |
| authorships[0].institutions[0].id | https://openalex.org/I20231570 |
| authorships[0].institutions[0].ror | https://ror.org/02v51f717 |
| authorships[0].institutions[0].type | education |
| authorships[0].institutions[0].lineage | https://openalex.org/I20231570 |
| authorships[0].institutions[0].country_code | CN |
| authorships[0].institutions[0].display_name | Peking University |
| authorships[0].institutions[1].id | https://openalex.org/I4210124809 |
| authorships[0].institutions[1].ror | https://ror.org/035adwg89 |
| authorships[0].institutions[1].type | healthcare |
| authorships[0].institutions[1].lineage | https://openalex.org/I4210124809 |
| authorships[0].institutions[1].country_code | CN |
| authorships[0].institutions[1].display_name | Peking University People's Hospital |
| authorships[0].author_position | first |
| authorships[0].raw_author_name | Linong Ji |
| authorships[0].is_corresponding | False |
| authorships[0].raw_affiliation_strings | Department of Endocrinology, Peking University People's Hospital, Beijing, China |
| authorships[1].author.id | https://openalex.org/A5109645255 |
| authorships[1].author.orcid | |
| authorships[1].author.display_name | Jianhua Ma |
| authorships[1].affiliations[0].raw_affiliation_string | Department of Endocrinology, Nanjing First Hospital, Nanjing, Jiangsu, China |
| authorships[1].author_position | middle |
| authorships[1].raw_author_name | Jianhua Ma |
| authorships[1].is_corresponding | False |
| authorships[1].raw_affiliation_strings | Department of Endocrinology, Nanjing First Hospital, Nanjing, Jiangsu, China |
| authorships[2].author.id | https://openalex.org/A5101893734 |
| authorships[2].author.orcid | https://orcid.org/0000-0003-2054-3398 |
| authorships[2].author.display_name | Yujin Ma |
| authorships[2].countries | CN |
| authorships[2].affiliations[0].institution_ids | https://openalex.org/I4210134906 |
| authorships[2].affiliations[0].raw_affiliation_string | Department of Endocrinology, The First Affiliated Hospital of Henan University of Science and Technology, Luoyang, Henan, China |
| authorships[2].institutions[0].id | https://openalex.org/I4210134906 |
| authorships[2].institutions[0].ror | https://ror.org/035zbbv42 |
| authorships[2].institutions[0].type | healthcare |
| authorships[2].institutions[0].lineage | https://openalex.org/I4210134906 |
| authorships[2].institutions[0].country_code | CN |
| authorships[2].institutions[0].display_name | First Affiliated Hospital of Henan University of Science and Technology |
| authorships[2].author_position | middle |
| authorships[2].raw_author_name | Yujin Ma |
| authorships[2].is_corresponding | False |
| authorships[2].raw_affiliation_strings | Department of Endocrinology, The First Affiliated Hospital of Henan University of Science and Technology, Luoyang, Henan, China |
| authorships[3].author.id | https://openalex.org/A5088900005 |
| authorships[3].author.orcid | https://orcid.org/0000-0003-0230-9258 |
| authorships[3].author.display_name | Zhifeng Cheng |
| authorships[3].countries | CN |
| authorships[3].affiliations[0].institution_ids | https://openalex.org/I156144747, https://openalex.org/I4210122309 |
| authorships[3].affiliations[0].raw_affiliation_string | Department of Endocrinology, The Fourth Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang, China |
| authorships[3].institutions[0].id | https://openalex.org/I4210122309 |
| authorships[3].institutions[0].ror | https://ror.org/02s7c9e98 |
| authorships[3].institutions[0].type | healthcare |
| authorships[3].institutions[0].lineage | https://openalex.org/I4210122309 |
| authorships[3].institutions[0].country_code | CN |
| authorships[3].institutions[0].display_name | Fourth Affiliated Hospital of Harbin Medical University |
| authorships[3].institutions[1].id | https://openalex.org/I156144747 |
| authorships[3].institutions[1].ror | https://ror.org/05jscf583 |
| authorships[3].institutions[1].type | education |
| authorships[3].institutions[1].lineage | https://openalex.org/I156144747 |
| authorships[3].institutions[1].country_code | CN |
| authorships[3].institutions[1].display_name | Harbin Medical University |
| authorships[3].author_position | middle |
| authorships[3].raw_author_name | Zhifeng Cheng |
| authorships[3].is_corresponding | False |
| authorships[3].raw_affiliation_strings | Department of Endocrinology, The Fourth Affiliated Hospital of Harbin Medical University, Harbin, Heilongjiang, China |
| authorships[4].author.id | https://openalex.org/A5058417870 |
| authorships[4].author.orcid | https://orcid.org/0000-0003-3005-183X |
| authorships[4].author.display_name | Shenglian Gan |
| authorships[4].countries | CN |
| authorships[4].affiliations[0].institution_ids | https://openalex.org/I4210123733 |
| authorships[4].affiliations[0].raw_affiliation_string | Department of Endocrinology, Changde First People's Hospital, Changde, Hunan, China |
| authorships[4].institutions[0].id | https://openalex.org/I4210123733 |
| authorships[4].institutions[0].ror | https://ror.org/02h2ywm64 |
| authorships[4].institutions[0].type | healthcare |
| authorships[4].institutions[0].lineage | https://openalex.org/I4210123733 |
| authorships[4].institutions[0].country_code | CN |
| authorships[4].institutions[0].display_name | The First People's Hospital of Changde |
| authorships[4].author_position | middle |
| authorships[4].raw_author_name | Shenglian Gan |
| authorships[4].is_corresponding | False |
| authorships[4].raw_affiliation_strings | Department of Endocrinology, Changde First People's Hospital, Changde, Hunan, China |
| authorships[5].author.id | https://openalex.org/A5066470511 |
| authorships[5].author.orcid | https://orcid.org/0000-0003-0822-6066 |
| authorships[5].author.display_name | Guoyue Yuan |
| authorships[5].countries | CN |
| authorships[5].affiliations[0].institution_ids | https://openalex.org/I115592961, https://openalex.org/I4210111628 |
| authorships[5].affiliations[0].raw_affiliation_string | Department of Endocrinology, Affiliated Hospital of Jiangsu University, Zhenjiang, Jiangsu, China |
| authorships[5].institutions[0].id | https://openalex.org/I4210111628 |
| authorships[5].institutions[0].ror | https://ror.org/028pgd321 |
| authorships[5].institutions[0].type | healthcare |
| authorships[5].institutions[0].lineage | https://openalex.org/I4210111628 |
| authorships[5].institutions[0].country_code | CN |
| authorships[5].institutions[0].display_name | Affiliated Hospital of Jiangsu University |
| authorships[5].institutions[1].id | https://openalex.org/I115592961 |
| authorships[5].institutions[1].ror | https://ror.org/03jc41j30 |
| authorships[5].institutions[1].type | education |
| authorships[5].institutions[1].lineage | https://openalex.org/I115592961 |
| authorships[5].institutions[1].country_code | CN |
| authorships[5].institutions[1].display_name | Jiangsu University |
| authorships[5].author_position | middle |
| authorships[5].raw_author_name | Guoyue Yuan |
| authorships[5].is_corresponding | False |
| authorships[5].raw_affiliation_strings | Department of Endocrinology, Affiliated Hospital of Jiangsu University, Zhenjiang, Jiangsu, China |
| authorships[6].author.id | https://openalex.org/A5102958910 |
| authorships[6].author.orcid | https://orcid.org/0000-0003-2048-7752 |
| authorships[6].author.display_name | Dexue Liu |
| authorships[6].countries | CN |
| authorships[6].affiliations[0].institution_ids | https://openalex.org/I4210135908 |
| authorships[6].affiliations[0].raw_affiliation_string | Department of Endocrinology, The First Affiliated Hospital of Nanyang Medical College, Nanyang, Henan, China |
| authorships[6].institutions[0].id | https://openalex.org/I4210135908 |
| authorships[6].institutions[0].ror | https://ror.org/03j450x81 |
| authorships[6].institutions[0].type | healthcare |
| authorships[6].institutions[0].lineage | https://openalex.org/I4210135908 |
| authorships[6].institutions[0].country_code | CN |
| authorships[6].institutions[0].display_name | Nanyang Medical College |
| authorships[6].author_position | middle |
| authorships[6].raw_author_name | Dexue Liu |
| authorships[6].is_corresponding | False |
| authorships[6].raw_affiliation_strings | Department of Endocrinology, The First Affiliated Hospital of Nanyang Medical College, Nanyang, Henan, China |
| authorships[7].author.id | https://openalex.org/A5042206062 |
| authorships[7].author.orcid | https://orcid.org/0000-0002-1546-8891 |
| authorships[7].author.display_name | Sheli Li |
| authorships[7].countries | CN |
| authorships[7].affiliations[0].institution_ids | https://openalex.org/I4210102412 |
| authorships[7].affiliations[0].raw_affiliation_string | Department of Endocrinology, Affiliated Hospital of Yan'an University, Yan'an, Shangxi, China |
| authorships[7].institutions[0].id | https://openalex.org/I4210102412 |
| authorships[7].institutions[0].ror | https://ror.org/01dyr7034 |
| authorships[7].institutions[0].type | education |
| authorships[7].institutions[0].lineage | https://openalex.org/I4210102412 |
| authorships[7].institutions[0].country_code | CN |
| authorships[7].institutions[0].display_name | Yan'an University |
| authorships[7].author_position | middle |
| authorships[7].raw_author_name | Sheli Li |
| authorships[7].is_corresponding | False |
| authorships[7].raw_affiliation_strings | Department of Endocrinology, Affiliated Hospital of Yan'an University, Yan'an, Shangxi, China |
| authorships[8].author.id | https://openalex.org/A5100345609 |
| authorships[8].author.orcid | https://orcid.org/0000-0001-6012-5305 |
| authorships[8].author.display_name | Yu Liu |
| authorships[8].countries | CN |
| authorships[8].affiliations[0].institution_ids | https://openalex.org/I83519826 |
| authorships[8].affiliations[0].raw_affiliation_string | Department of Endocrinology, Nanjing Medical University Hospital, Nanjing, Jiangsu, China |
| authorships[8].institutions[0].id | https://openalex.org/I83519826 |
| authorships[8].institutions[0].ror | https://ror.org/059gcgy73 |
| authorships[8].institutions[0].type | education |
| authorships[8].institutions[0].lineage | https://openalex.org/I83519826 |
| authorships[8].institutions[0].country_code | CN |
| authorships[8].institutions[0].display_name | Nanjing Medical University |
| authorships[8].author_position | middle |
| authorships[8].raw_author_name | Yu Liu |
| authorships[8].is_corresponding | False |
| authorships[8].raw_affiliation_strings | Department of Endocrinology, Nanjing Medical University Hospital, Nanjing, Jiangsu, China |
| authorships[9].author.id | https://openalex.org/A5063891857 |
| authorships[9].author.orcid | https://orcid.org/0000-0002-5783-1281 |
| authorships[9].author.display_name | Xia Xue |
| authorships[9].countries | CN |
| authorships[9].affiliations[0].institution_ids | https://openalex.org/I4210106451 |
| authorships[9].affiliations[0].raw_affiliation_string | Department of Endocrinology, Jinan Central Hospital, Jinan, Shandong, China |
| authorships[9].institutions[0].id | https://openalex.org/I4210106451 |
| authorships[9].institutions[0].ror | https://ror.org/01fr19c68 |
| authorships[9].institutions[0].type | healthcare |
| authorships[9].institutions[0].lineage | https://openalex.org/I4210106451 |
| authorships[9].institutions[0].country_code | CN |
| authorships[9].institutions[0].display_name | Jinan Central Hospital |
| authorships[9].author_position | middle |
| authorships[9].raw_author_name | Xia Xue |
| authorships[9].is_corresponding | False |
| authorships[9].raw_affiliation_strings | Department of Endocrinology, Jinan Central Hospital, Jinan, Shandong, China |
| authorships[10].author.id | https://openalex.org/A5088843765 |
| authorships[10].author.orcid | https://orcid.org/0000-0002-7493-8347 |
| authorships[10].author.display_name | Jie Bai |
| authorships[10].countries | CN |
| authorships[10].affiliations[0].institution_ids | https://openalex.org/I4210160222 |
| authorships[10].affiliations[0].raw_affiliation_string | Department of Endocrinology, Liaocheng People's Hospital, Liaocheng, Shandong, China |
| authorships[10].institutions[0].id | https://openalex.org/I4210160222 |
| authorships[10].institutions[0].ror | https://ror.org/052vn2478 |
| authorships[10].institutions[0].type | healthcare |
| authorships[10].institutions[0].lineage | https://openalex.org/I4210160222 |
| authorships[10].institutions[0].country_code | CN |
| authorships[10].institutions[0].display_name | Liaocheng People's Hospital |
| authorships[10].author_position | middle |
| authorships[10].raw_author_name | Jie Bai |
| authorships[10].is_corresponding | False |
| authorships[10].raw_affiliation_strings | Department of Endocrinology, Liaocheng People's Hospital, Liaocheng, Shandong, China |
| authorships[11].author.id | https://openalex.org/A5100366709 |
| authorships[11].author.orcid | https://orcid.org/0000-0003-1142-0900 |
| authorships[11].author.display_name | Kun Wang |
| authorships[11].countries | CN |
| authorships[11].affiliations[0].institution_ids | https://openalex.org/I4210156162 |
| authorships[11].affiliations[0].raw_affiliation_string | Department of Endocrinology, Nanjing Jiangning Hospital, Nanjing, Jiangsu, China |
| authorships[11].institutions[0].id | https://openalex.org/I4210156162 |
| authorships[11].institutions[0].ror | https://ror.org/04sk80178 |
| authorships[11].institutions[0].type | healthcare |
| authorships[11].institutions[0].lineage | https://openalex.org/I4210156162 |
| authorships[11].institutions[0].country_code | CN |
| authorships[11].institutions[0].display_name | Nanjing Jiangning Hospital |
| authorships[11].author_position | middle |
| authorships[11].raw_author_name | Kun Wang |
| authorships[11].is_corresponding | False |
| authorships[11].raw_affiliation_strings | Department of Endocrinology, Nanjing Jiangning Hospital, Nanjing, Jiangsu, China |
| authorships[12].author.id | https://openalex.org/A5083527446 |
| authorships[12].author.orcid | https://orcid.org/0000-0002-9040-9103 |
| authorships[12].author.display_name | Hanqing Cai |
| authorships[12].countries | CN |
| authorships[12].affiliations[0].institution_ids | https://openalex.org/I4210142484 |
| authorships[12].affiliations[0].raw_affiliation_string | Department of Endocrinology, The Second Hospital of Jilin University, Changchun, Jilin, China |
| authorships[12].institutions[0].id | https://openalex.org/I4210142484 |
| authorships[12].institutions[0].ror | https://ror.org/03x6hbh34 |
| authorships[12].institutions[0].type | healthcare |
| authorships[12].institutions[0].lineage | https://openalex.org/I4210142484 |
| authorships[12].institutions[0].country_code | CN |
| authorships[12].institutions[0].display_name | Second Affiliated Hospital of Jilin University |
| authorships[12].author_position | middle |
| authorships[12].raw_author_name | Hanqing Cai |
| authorships[12].is_corresponding | False |
| authorships[12].raw_affiliation_strings | Department of Endocrinology, The Second Hospital of Jilin University, Changchun, Jilin, China |
| authorships[13].author.id | https://openalex.org/A5100404332 |
| authorships[13].author.orcid | https://orcid.org/0000-0003-0350-0353 |
| authorships[13].author.display_name | Shu Li |
| authorships[13].countries | CN |
| authorships[13].affiliations[0].institution_ids | https://openalex.org/I4210138268 |
| authorships[13].affiliations[0].raw_affiliation_string | Department of Endocrinology, Huizhou Central People's Hospital, Huizhou, Guangdong, China |
| authorships[13].institutions[0].id | https://openalex.org/I4210138268 |
| authorships[13].institutions[0].ror | https://ror.org/04bwajd86 |
| authorships[13].institutions[0].type | healthcare |
| authorships[13].institutions[0].lineage | https://openalex.org/I4210138268 |
| authorships[13].institutions[0].country_code | CN |
| authorships[13].institutions[0].display_name | Huizhou Central People's Hospital |
| authorships[13].author_position | middle |
| authorships[13].raw_author_name | Shu Li |
| authorships[13].is_corresponding | False |
| authorships[13].raw_affiliation_strings | Department of Endocrinology, Huizhou Central People's Hospital, Huizhou, Guangdong, China |
| authorships[14].author.id | https://openalex.org/A5100374028 |
| authorships[14].author.orcid | https://orcid.org/0009-0008-9063-6921 |
| authorships[14].author.display_name | Kui Liu |
| authorships[14].affiliations[0].raw_affiliation_string | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| authorships[14].author_position | middle |
| authorships[14].raw_author_name | Kui Liu |
| authorships[14].is_corresponding | False |
| authorships[14].raw_affiliation_strings | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| authorships[15].author.id | https://openalex.org/A5100519050 |
| authorships[15].author.orcid | |
| authorships[15].author.display_name | Meng Yu |
| authorships[15].affiliations[0].raw_affiliation_string | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| authorships[15].author_position | middle |
| authorships[15].raw_author_name | Meng Yu |
| authorships[15].is_corresponding | False |
| authorships[15].raw_affiliation_strings | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| authorships[16].author.id | https://openalex.org/A5100352454 |
| authorships[16].author.orcid | https://orcid.org/0000-0003-2943-055X |
| authorships[16].author.display_name | Liping Liu |
| authorships[16].affiliations[0].raw_affiliation_string | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| authorships[16].author_position | last |
| authorships[16].raw_author_name | Liping Liu |
| authorships[16].is_corresponding | True |
| authorships[16].raw_affiliation_strings | Shenzhen HighTide Biopharmaceutical Ltd, Shenzhen, Guangdong, China |
| has_content.pdf | True |
| has_content.grobid_xml | False |
| is_paratext | False |
| open_access.is_oa | True |
| open_access.oa_url | https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdf |
| open_access.oa_status | gold |
| open_access.any_repository_has_fulltext | False |
| created_date | 2025-03-03T00:00:00 |
| display_name | Berberine Ursodeoxycholate for the Treatment of Type 2 Diabetes |
| has_fulltext | False |
| is_retracted | False |
| updated_date | 2025-11-06T03:46:38.306776 |
| primary_topic.id | https://openalex.org/T12059 |
| primary_topic.field.id | https://openalex.org/fields/27 |
| primary_topic.field.display_name | Medicine |
| primary_topic.score | 0.9997000098228455 |
| primary_topic.domain.id | https://openalex.org/domains/4 |
| primary_topic.domain.display_name | Health Sciences |
| primary_topic.subfield.id | https://openalex.org/subfields/2736 |
| primary_topic.subfield.display_name | Pharmacology |
| primary_topic.display_name | Berberine and alkaloids research |
| related_works | https://openalex.org/W1859326399, https://openalex.org/W204855368, https://openalex.org/W1966127473, https://openalex.org/W2085650940, https://openalex.org/W4390338738, https://openalex.org/W2169836118, https://openalex.org/W2081991953, https://openalex.org/W3033200810, https://openalex.org/W4309072394, https://openalex.org/W3128414331 |
| cited_by_count | 11 |
| counts_by_year[0].year | 2025 |
| counts_by_year[0].cited_by_count | 11 |
| locations_count | 3 |
| best_oa_location.id | doi:10.1001/jamanetworkopen.2024.62185 |
| best_oa_location.is_oa | True |
| best_oa_location.source.id | https://openalex.org/S4210217848 |
| best_oa_location.source.issn | 2574-3805 |
| best_oa_location.source.type | journal |
| best_oa_location.source.is_oa | True |
| best_oa_location.source.issn_l | 2574-3805 |
| best_oa_location.source.is_core | True |
| best_oa_location.source.is_in_doaj | True |
| best_oa_location.source.display_name | JAMA Network Open |
| best_oa_location.source.host_organization | https://openalex.org/P4310320259 |
| best_oa_location.source.host_organization_name | American Medical Association |
| best_oa_location.source.host_organization_lineage | https://openalex.org/P4310320259 |
| best_oa_location.license | |
| best_oa_location.pdf_url | https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdf |
| best_oa_location.version | publishedVersion |
| best_oa_location.raw_type | journal-article |
| best_oa_location.license_id | |
| best_oa_location.is_accepted | True |
| best_oa_location.is_published | True |
| best_oa_location.raw_source_name | JAMA Network Open |
| best_oa_location.landing_page_url | https://doi.org/10.1001/jamanetworkopen.2024.62185 |
| primary_location.id | doi:10.1001/jamanetworkopen.2024.62185 |
| primary_location.is_oa | True |
| primary_location.source.id | https://openalex.org/S4210217848 |
| primary_location.source.issn | 2574-3805 |
| primary_location.source.type | journal |
| primary_location.source.is_oa | True |
| primary_location.source.issn_l | 2574-3805 |
| primary_location.source.is_core | True |
| primary_location.source.is_in_doaj | True |
| primary_location.source.display_name | JAMA Network Open |
| primary_location.source.host_organization | https://openalex.org/P4310320259 |
| primary_location.source.host_organization_name | American Medical Association |
| primary_location.source.host_organization_lineage | https://openalex.org/P4310320259 |
| primary_location.license | |
| primary_location.pdf_url | https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2830820/ji_2025_oi_241733_1740068767.56731.pdf |
| primary_location.version | publishedVersion |
| primary_location.raw_type | journal-article |
| primary_location.license_id | |
| primary_location.is_accepted | True |
| primary_location.is_published | True |
| primary_location.raw_source_name | JAMA Network Open |
| primary_location.landing_page_url | https://doi.org/10.1001/jamanetworkopen.2024.62185 |
| publication_date | 2025-03-03 |
| publication_year | 2025 |
| referenced_works | https://openalex.org/W44929223, https://openalex.org/W3020528808, https://openalex.org/W3081655479, https://openalex.org/W3131356637, https://openalex.org/W2887185538, https://openalex.org/W4283814486, https://openalex.org/W3201436860, https://openalex.org/W4399560402, https://openalex.org/W4292528167, https://openalex.org/W1981066923, https://openalex.org/W2137069992, https://openalex.org/W2160234571, https://openalex.org/W2068398835, https://openalex.org/W4393857320, https://openalex.org/W2129284446, https://openalex.org/W2462514513, https://openalex.org/W4200619278, https://openalex.org/W3010858550, https://openalex.org/W2137573856, https://openalex.org/W2904952494, https://openalex.org/W2005448673, https://openalex.org/W2084686255, https://openalex.org/W2036462559, https://openalex.org/W2160835284, https://openalex.org/W3010351291, https://openalex.org/W4379093314, https://openalex.org/W2955615964, https://openalex.org/W2979736764, https://openalex.org/W3160407865, https://openalex.org/W2095869629, https://openalex.org/W2166169943, https://openalex.org/W4308367810, https://openalex.org/W3099525102, https://openalex.org/W2789732774, https://openalex.org/W4406660574, https://openalex.org/W2607031541 |
| referenced_works_count | 36 |
| abstract_inverted_index.) | 106 |
| abstract_inverted_index.2 | 8, 69 |
| abstract_inverted_index.3 | 491 |
| abstract_inverted_index.8 | 91 |
| abstract_inverted_index.= | 132, 140, 149, 327 |
| abstract_inverted_index.A | 102 |
| abstract_inverted_index.P | 326, 340 |
| abstract_inverted_index.a | 23, 100, 114, 186, 427 |
| abstract_inverted_index.(n | 131, 139, 148 |
| abstract_inverted_index.12 | 318 |
| abstract_inverted_index.1c | 103, 105, 162, 195, 248, 296, 314, 353, 467 |
| abstract_inverted_index.59 | 416 |
| abstract_inverted_index.72 | 235 |
| abstract_inverted_index.In | 451 |
| abstract_inverted_index.No | 440 |
| abstract_inverted_index.To | 43 |
| abstract_inverted_index.an | 445, 499 |
| abstract_inverted_index.as | 200, 259 |
| abstract_inverted_index.at | 316 |
| abstract_inverted_index.by | 264, 496 |
| abstract_inverted_index.in | 51, 77, 261, 266, 293, 298, 311, 362, 366, 385, 392, 415, 461, 464, 471, 488 |
| abstract_inverted_index.is | 40, 56 |
| abstract_inverted_index.mg | 136, 145 |
| abstract_inverted_index.of | 2, 37, 49, 95, 108, 389, 431 |
| abstract_inverted_index.or | 92 |
| abstract_inverted_index.to | 32, 110, 129, 167, 324, 338, 438, 444 |
| abstract_inverted_index.(in | 422 |
| abstract_inverted_index.110 | 403 |
| abstract_inverted_index.113 | 225 |
| abstract_inverted_index.12. | 169 |
| abstract_inverted_index.500 | 135 |
| abstract_inverted_index.CI, | 322, 336 |
| abstract_inverted_index.FPG | 270, 364 |
| abstract_inverted_index.Few | 1 |
| abstract_inverted_index.HbA | 161, 194, 247, 295, 313, 352, 466 |
| abstract_inverted_index.One | 420 |
| abstract_inverted_index.T2D | 54, 88, 228, 504 |
| abstract_inverted_index.The | 155, 179, 222, 283, 307, 493 |
| abstract_inverted_index.and | 47, 61, 65, 82, 97, 112, 142, 153, 176, 209, 214, 269, 333, 374, 387, 399, 449, 469, 474, 480, 505 |
| abstract_inverted_index.are | 485 |
| abstract_inverted_index.due | 443 |
| abstract_inverted_index.end | 157, 171, 181, 285 |
| abstract_inverted_index.for | 6, 189, 329, 343, 503 |
| abstract_inverted_index.had | 99, 113 |
| abstract_inverted_index.has | 29 |
| abstract_inverted_index.its | 506 |
| abstract_inverted_index.key | 472 |
| abstract_inverted_index.the | 3, 30, 34, 45, 160, 193, 201, 244, 294, 304, 312, 330, 344, 349, 363, 368, 375, 393, 407, 423, 465 |
| abstract_inverted_index.was | 159, 183, 250, 278, 287, 319, 397, 435, 478 |
| abstract_inverted_index.who | 89, 238 |
| abstract_inverted_index.(95% | 321, 335 |
| abstract_inverted_index.(HbA | 104 |
| abstract_inverted_index.(SD) | 246, 360 |
| abstract_inverted_index..04) | 328 |
| abstract_inverted_index.1000 | 144 |
| abstract_inverted_index.2022 | 81 |
| abstract_inverted_index.25.5 | 256 |
| abstract_inverted_index.37), | 141 |
| abstract_inverted_index.38), | 133 |
| abstract_inverted_index.38). | 150 |
| abstract_inverted_index.54.3 | 232 |
| abstract_inverted_index.7.0% | 109 |
| abstract_inverted_index.8.2% | 251 |
| abstract_inverted_index.Main | 151 |
| abstract_inverted_index.This | 67 |
| abstract_inverted_index.[SD] | 230 |
| abstract_inverted_index.age, | 231 |
| abstract_inverted_index.body | 253 |
| abstract_inverted_index.both | 299, 367 |
| abstract_inverted_index.core | 35 |
| abstract_inverted_index.diet | 60, 96 |
| abstract_inverted_index.from | 165, 198 |
| abstract_inverted_index.less | 120 |
| abstract_inverted_index.male | 236 |
| abstract_inverted_index.mass | 254 |
| abstract_inverted_index.mean | 245, 309, 359 |
| abstract_inverted_index.more | 93 |
| abstract_inverted_index.oral | 500 |
| abstract_inverted_index.safe | 398, 479 |
| abstract_inverted_index.than | 121 |
| abstract_inverted_index.that | 55 |
| abstract_inverted_index.this | 452 |
| abstract_inverted_index.time | 207, 216 |
| abstract_inverted_index.type | 7 |
| abstract_inverted_index.week | 168, 317 |
| abstract_inverted_index.well | 400, 481 |
| abstract_inverted_index.were | 126, 218, 239, 356, 383 |
| abstract_inverted_index.with | 53, 59, 87, 192, 227, 289, 303, 348, 358, 402, 458 |
| abstract_inverted_index.(FPG) | 118 |
| abstract_inverted_index.(T2D) | 10 |
| abstract_inverted_index.(mean | 229 |
| abstract_inverted_index..001) | 342 |
| abstract_inverted_index.160.7 | 272 |
| abstract_inverted_index.1:1:1 | 128 |
| abstract_inverted_index.2023, | 84 |
| abstract_inverted_index.250.5 | 122 |
| abstract_inverted_index.Among | 241 |
| abstract_inverted_index.China | 78 |
| abstract_inverted_index.March | 80 |
| abstract_inverted_index.These | 483 |
| abstract_inverted_index.Trial | 508 |
| abstract_inverted_index.being | 486 |
| abstract_inverted_index.daily | 138, 147 |
| abstract_inverted_index.event | 430 |
| abstract_inverted_index.group | 213, 332, 346, 370, 377 |
| abstract_inverted_index.level | 107, 119, 163, 196, 249, 297, 315, 354, 365, 468 |
| abstract_inverted_index.liver | 390, 475 |
| abstract_inverted_index.mild, | 413 |
| abstract_inverted_index.model | 188 |
| abstract_inverted_index.phase | 68, 490 |
| abstract_inverted_index.point | 158, 182, 217, 286 |
| abstract_inverted_index.study | 223 |
| abstract_inverted_index.these | 242 |
| abstract_inverted_index.treat | 33 |
| abstract_inverted_index.twice | 137, 146 |
| abstract_inverted_index.using | 185 |
| abstract_inverted_index.weeks | 94 |
| abstract_inverted_index.which | 434 |
| abstract_inverted_index.(3.7), | 257 |
| abstract_inverted_index.(38.3) | 273 |
| abstract_inverted_index.10.5%, | 111 |
| abstract_inverted_index.500-mg | 331, 369, 424 |
| abstract_inverted_index.[10.6] | 233 |
| abstract_inverted_index.[21.8] | 379 |
| abstract_inverted_index.[38.2] | 372 |
| abstract_inverted_index.across | 280 |
| abstract_inverted_index.assess | 44 |
| abstract_inverted_index.beyond | 15 |
| abstract_inverted_index.burden | 14 |
| abstract_inverted_index.change | 164, 197 |
| abstract_inverted_index.event. | 447 |
| abstract_inverted_index.group) | 425 |
| abstract_inverted_index.group, | 205 |
| abstract_inverted_index.group. | 306, 351, 395 |
| abstract_inverted_index.groups | 301 |
| abstract_inverted_index.height | 265 |
| abstract_inverted_index.index, | 255 |
| abstract_inverted_index.injury | 391 |
| abstract_inverted_index.level, | 271 |
| abstract_inverted_index.lipids | 386 |
| abstract_inverted_index.meters | 267 |
| abstract_inverted_index.mg/dL) | 373, 380 |
| abstract_inverted_index.mg/dL. | 123, 274 |
| abstract_inverted_index.option | 502 |
| abstract_inverted_index.plasma | 116 |
| abstract_inverted_index.point, | 208 |
| abstract_inverted_index.points | 172 |
| abstract_inverted_index.safety | 46 |
| abstract_inverted_index.study. | 408 |
| abstract_inverted_index.trial, | 75, 456 |
| abstract_inverted_index.weight | 260 |
| abstract_inverted_index.years; | 234 |
| abstract_inverted_index.(0.8%); | 252 |
| abstract_inverted_index.(52.2%) | 418 |
| abstract_inverted_index.(97.3%) | 405 |
| abstract_inverted_index.1000-mg | 345, 376, 394 |
| abstract_inverted_index.12-week | 72 |
| abstract_inverted_index.Design, | 63 |
| abstract_inverted_index.HTD1801 | 50, 134, 143, 300, 396, 459, 477, 497 |
| abstract_inverted_index.January | 83 |
| abstract_inverted_index.Results | 221 |
| abstract_inverted_index.address | 12 |
| abstract_inverted_index.adverse | 410, 429, 446 |
| abstract_inverted_index.aspects | 36 |
| abstract_inverted_index.between | 79, 211 |
| abstract_inverted_index.disease | 13, 39, 276 |
| abstract_inverted_index.divided | 263 |
| abstract_inverted_index.effects | 494 |
| abstract_inverted_index.events, | 411 |
| abstract_inverted_index.fasting | 115 |
| abstract_inverted_index.glucose | 117 |
| abstract_inverted_index.groups. | 282, 381 |
| abstract_inverted_index.markers | 388 |
| abstract_inverted_index.ongoing | 489 |
| abstract_inverted_index.patient | 421 |
| abstract_inverted_index.placebo | 130, 305, 350 |
| abstract_inverted_index.primary | 156, 180, 284 |
| abstract_inverted_index.related | 437 |
| abstract_inverted_index.retinal | 432 |
| abstract_inverted_index.serious | 428 |
| abstract_inverted_index.support | 498 |
| abstract_inverted_index.whether | 19 |
| abstract_inverted_index.−0.4% | 320 |
| abstract_inverted_index.−0.7% | 334 |
| abstract_inverted_index.(−13.0 | 371 |
| abstract_inverted_index.(−18.4 | 378 |
| abstract_inverted_index.Baseline | 275 |
| abstract_inverted_index.Measures | 154 |
| abstract_inverted_index.Outcomes | 152 |
| abstract_inverted_index.Patients | 125 |
| abstract_inverted_index.Setting, | 64 |
| abstract_inverted_index.[63.7%]) | 237 |
| abstract_inverted_index.achieved | 288 |
| abstract_inverted_index.analyzed | 184 |
| abstract_inverted_index.balanced | 279 |
| abstract_inverted_index.baseline | 166, 199 |
| abstract_inverted_index.clinical | 74, 455 |
| abstract_inverted_index.compared | 302, 347 |
| abstract_inverted_index.control. | 17 |
| abstract_inverted_index.diabetes | 9 |
| abstract_inverted_index.efficacy | 48 |
| abstract_inverted_index.findings | 484 |
| abstract_inverted_index.glycemic | 16 |
| abstract_inverted_index.hepatic, | 175 |
| abstract_inverted_index.included | 85, 173, 224 |
| abstract_inverted_index.observed | 384 |
| abstract_inverted_index.occurred | 414 |
| abstract_inverted_index.overall. | 419 |
| abstract_inverted_index.patients | 52, 86, 226, 404, 417, 441 |
| abstract_inverted_index.repeated | 190 |
| abstract_inverted_index.resulted | 460 |
| abstract_inverted_index.severity | 277 |
| abstract_inverted_index.squared; | 268 |
| abstract_inverted_index.studies. | 492 |
| abstract_inverted_index.unlikely | 436 |
| abstract_inverted_index.−0.79% | 323 |
| abstract_inverted_index.−1.10% | 337 |
| abstract_inverted_index.Examining | 18 |
| abstract_inverted_index.Objective | 42 |
| abstract_inverted_index.Relevance | 450 |
| abstract_inverted_index.Secondary | 170 |
| abstract_inverted_index.Treatment | 204 |
| abstract_inverted_index.available | 4 |
| abstract_inverted_index.berberine | 20 |
| abstract_inverted_index.conducted | 76 |
| abstract_inverted_index.confirmed | 487 |
| abstract_inverted_index.dependent | 202 |
| abstract_inverted_index.exercise, | 98 |
| abstract_inverted_index.exercise. | 62 |
| abstract_inverted_index.generally | 412 |
| abstract_inverted_index.glycemic, | 174 |
| abstract_inverted_index.gut-liver | 25 |
| abstract_inverted_index.kilograms | 262 |
| abstract_inverted_index.measures, | 191 |
| abstract_inverted_index.metabolic | 27, 38 |
| abstract_inverted_index.patients, | 243 |
| abstract_inverted_index.potential | 31 |
| abstract_inverted_index.therapies | 5 |
| abstract_inverted_index.treatment | 212, 281, 457, 501 |
| abstract_inverted_index.underwent | 90 |
| abstract_inverted_index.variable. | 203 |
| abstract_inverted_index.−0.03%; | 325 |
| abstract_inverted_index.−0.35%; | 339 |
| abstract_inverted_index.(HTD1801), | 22 |
| abstract_inverted_index.Importance | 0 |
| abstract_inverted_index.Reductions | 382 |
| abstract_inverted_index.calculated | 258 |
| abstract_inverted_index.completing | 406 |
| abstract_inverted_index.controlled | 58 |
| abstract_inverted_index.difference | 310 |
| abstract_inverted_index.hemoglobin | 101 |
| abstract_inverted_index.important. | 41 |
| abstract_inverted_index.modulator, | 28 |
| abstract_inverted_index.paralleled | 357 |
| abstract_inverted_index.randomized | 73, 127, 454 |
| abstract_inverted_index.reductions | 292, 355, 463 |
| abstract_inverted_index.tolerated, | 401 |
| abstract_inverted_index.tolerated. | 482 |
| abstract_inverted_index.treatment. | 439 |
| abstract_inverted_index.variables. | 220 |
| abstract_inverted_index.Conclusions | 448 |
| abstract_inverted_index.Identifier: | 511 |
| abstract_inverted_index.NCT06411275 | 512 |
| abstract_inverted_index.experienced | 426 |
| abstract_inverted_index.hemorrhage, | 433 |
| abstract_inverted_index.independent | 219 |
| abstract_inverted_index.interaction | 210 |
| abstract_inverted_index.measurement | 206, 215 |
| abstract_inverted_index.parameters. | 178, 476 |
| abstract_inverted_index.randomized. | 240 |
| abstract_inverted_index.significant | 290, 462 |
| abstract_inverted_index.&amp;lt; | 341 |
| abstract_inverted_index.Participants | 66 |
| abstract_inverted_index.Registration | 509 |
| abstract_inverted_index.demonstrated | 495 |
| abstract_inverted_index.discontinued | 442 |
| abstract_inverted_index.improvements | 361, 470 |
| abstract_inverted_index.inadequately | 57 |
| abstract_inverted_index.Interventions | 124 |
| abstract_inverted_index.double-blind, | 70 |
| abstract_inverted_index.least-squares | 308 |
| abstract_inverted_index.mixed-effects | 187 |
| abstract_inverted_index.comorbidities. | 507 |
| abstract_inverted_index.dose-dependent | 291 |
| abstract_inverted_index.first-in-class | 24 |
| abstract_inverted_index.cardiometabolic | 177, 473 |
| abstract_inverted_index.comprehensively | 11 |
| abstract_inverted_index.ursodeoxycholate | 21 |
| abstract_inverted_index.anti-inflammatory | 26 |
| abstract_inverted_index.ClinicalTrials.gov | 510 |
| abstract_inverted_index.Treatment-emergent | 409 |
| abstract_inverted_index.placebo-controlled | 453 |
| abstract_inverted_index.placebo-controlled, | 71 |
| cited_by_percentile_year.max | 99 |
| cited_by_percentile_year.min | 98 |
| corresponding_author_ids | https://openalex.org/A5100352454 |
| countries_distinct_count | 1 |
| institutions_distinct_count | 17 |
| sustainable_development_goals[0].id | https://metadata.un.org/sdg/3 |
| sustainable_development_goals[0].score | 0.8500000238418579 |
| sustainable_development_goals[0].display_name | Good health and well-being |
| citation_normalized_percentile.value | 0.9979775 |
| citation_normalized_percentile.is_in_top_1_percent | True |
| citation_normalized_percentile.is_in_top_10_percent | True |