Treating Acute EXacerbation of COPD with Chinese HerbAL MedIcine to aid AntiBiotic Use Reduction (EXCALIBUR): study protocol of a randomised double-blind, placebo-controlled feasibility trial Article Swipe
 YOU?
·
· 2022
· Open Access
·
· DOI: https://doi.org/10.21203/rs.3.rs-1918642/v1
YOU?
·
· 2022
· Open Access
·
· DOI: https://doi.org/10.21203/rs.3.rs-1918642/v1
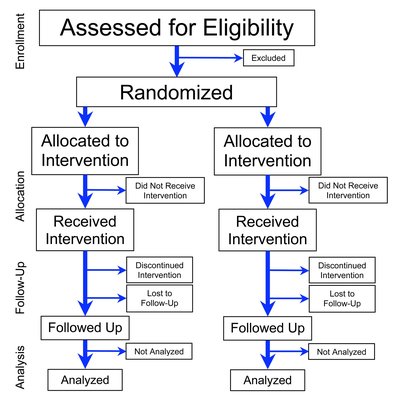
Background: Acute exacerbation of chronic obstructive pulmonary disease (AECOPD) are a major reason for consultations in primary care, hospital admissions, deterioration in function, and mortality. 70% of patients presenting with AECOPD in UK primary care are currently in receipt of antibiotics which is part of the standard care. However, the majority of exacerbations are not caused by bacteria. Finding effective non-antibiotic treatments for COPD exacerbations is a priority to reduce antibiotic use. The Chinese herbal medicine Shufeng Jieduâ (SFJD) has the potential to reduce treatment failure and duration of hospital stay. This study aims to test the feasibility of a randomised, double blind, placebo-controlled clinical trial on SFJD in AECOPD. Methods: This study is a phase III, two-arm individually double blind, randomised, placebo-controlled feasibility trial with nested qualitative study, coordinated by the Southampton clinical trial unit (SCTU). Patients aged ≥40 years old, with a current AECOPD, presenting with increased sputum purulence/ volume, or breathlessness, and for whom the GP is considering use of antibiotics, will be eligible to participate. We aim to recruit seven eligible participants per month, randomise them to receive either the patent Chinese herbal medicine SFJD capsules or placebo for 14 consecutive days and to follow up for 12 weeks. The primary outcomes include the feasibility of recruitment, study retention, and the completion of trial diaries. Discussion: If this trial demonstrates the feasibility of recruitment, delivery and follow-up, we will seek funding for a fully powered placebo-controlled trial of SFJD for the treatment of AECOPD in primary care. Trial registration : This trial is registered via ISRCTN on 1 st July 2021, identifier: ISRCTN26614726.
Related Topics
- Type
- preprint
- Language
- en
- Landing Page
- https://doi.org/10.21203/rs.3.rs-1918642/v1
- https://www.researchsquare.com/article/rs-1918642/latest.pdf
- OA Status
- green
- References
- 20
- Related Works
- 10
- OpenAlex ID
- https://openalex.org/W4293582268
Raw OpenAlex JSON
- OpenAlex ID
-
https://openalex.org/W4293582268Canonical identifier for this work in OpenAlex
- DOI
-
https://doi.org/10.21203/rs.3.rs-1918642/v1Digital Object Identifier
- Title
-
Treating Acute EXacerbation of COPD with Chinese HerbAL MedIcine to aid AntiBiotic Use Reduction (EXCALIBUR): study protocol of a randomised double-blind, placebo-controlled feasibility trialWork title
- Type
-
preprintOpenAlex work type
- Language
-
enPrimary language
- Publication year
-
2022Year of publication
- Publication date
-
2022-08-30Full publication date if available
- Authors
-
Xiao-Yang Hu, Tom Oliver, Merlin Willcox, Catherine Simpson, Kerensa Thorne, Jeanne Trill, Nick Francis, Beth Stuart, Mike Thomas, Paul Little, Jianping Liu, Gareth Griffiths, Michael MooreList of authors in order
- Landing page
-
https://doi.org/10.21203/rs.3.rs-1918642/v1Publisher landing page
- PDF URL
-
https://www.researchsquare.com/article/rs-1918642/latest.pdfDirect link to full text PDF
- Open access
-
YesWhether a free full text is available
- OA status
-
greenOpen access status per OpenAlex
- OA URL
-
https://www.researchsquare.com/article/rs-1918642/latest.pdfDirect OA link when available
- Concepts
-
Medicine, Exacerbation, Placebo, COPD, Randomized controlled trial, Clinical trial, Acute exacerbation of chronic obstructive pulmonary disease, Intensive care medicine, Physical therapy, Internal medicine, Alternative medicine, PathologyTop concepts (fields/topics) attached by OpenAlex
- Cited by
-
0Total citation count in OpenAlex
- References (count)
-
20Number of works referenced by this work
- Related works (count)
-
10Other works algorithmically related by OpenAlex
Full payload
| id | https://openalex.org/W4293582268 |
|---|---|
| doi | https://doi.org/10.21203/rs.3.rs-1918642/v1 |
| ids.doi | https://doi.org/10.21203/rs.3.rs-1918642/v1 |
| ids.openalex | https://openalex.org/W4293582268 |
| fwci | 0.0 |
| type | preprint |
| title | Treating Acute EXacerbation of COPD with Chinese HerbAL MedIcine to aid AntiBiotic Use Reduction (EXCALIBUR): study protocol of a randomised double-blind, placebo-controlled feasibility trial |
| biblio.issue | |
| biblio.volume | |
| biblio.last_page | |
| biblio.first_page | |
| topics[0].id | https://openalex.org/T10143 |
| topics[0].field.id | https://openalex.org/fields/27 |
| topics[0].field.display_name | Medicine |
| topics[0].score | 0.9979000091552734 |
| topics[0].domain.id | https://openalex.org/domains/4 |
| topics[0].domain.display_name | Health Sciences |
| topics[0].subfield.id | https://openalex.org/subfields/2740 |
| topics[0].subfield.display_name | Pulmonary and Respiratory Medicine |
| topics[0].display_name | Chronic Obstructive Pulmonary Disease (COPD) Research |
| topics[1].id | https://openalex.org/T13770 |
| topics[1].field.id | https://openalex.org/fields/36 |
| topics[1].field.display_name | Health Professions |
| topics[1].score | 0.9829999804496765 |
| topics[1].domain.id | https://openalex.org/domains/4 |
| topics[1].domain.display_name | Health Sciences |
| topics[1].subfield.id | https://openalex.org/subfields/3604 |
| topics[1].subfield.display_name | Emergency Medical Services |
| topics[1].display_name | Pediatric health and respiratory diseases |
| topics[2].id | https://openalex.org/T12418 |
| topics[2].field.id | https://openalex.org/fields/27 |
| topics[2].field.display_name | Medicine |
| topics[2].score | 0.9484999775886536 |
| topics[2].domain.id | https://openalex.org/domains/4 |
| topics[2].domain.display_name | Health Sciences |
| topics[2].subfield.id | https://openalex.org/subfields/2740 |
| topics[2].subfield.display_name | Pulmonary and Respiratory Medicine |
| topics[2].display_name | Respiratory and Cough-Related Research |
| is_xpac | False |
| apc_list | |
| apc_paid | |
| concepts[0].id | https://openalex.org/C71924100 |
| concepts[0].level | 0 |
| concepts[0].score | 0.9346504211425781 |
| concepts[0].wikidata | https://www.wikidata.org/wiki/Q11190 |
| concepts[0].display_name | Medicine |
| concepts[1].id | https://openalex.org/C2777014857 |
| concepts[1].level | 2 |
| concepts[1].score | 0.7290292978286743 |
| concepts[1].wikidata | https://www.wikidata.org/wiki/Q1383410 |
| concepts[1].display_name | Exacerbation |
| concepts[2].id | https://openalex.org/C27081682 |
| concepts[2].level | 3 |
| concepts[2].score | 0.7018404006958008 |
| concepts[2].wikidata | https://www.wikidata.org/wiki/Q269829 |
| concepts[2].display_name | Placebo |
| concepts[3].id | https://openalex.org/C2776780178 |
| concepts[3].level | 2 |
| concepts[3].score | 0.6238923668861389 |
| concepts[3].wikidata | https://www.wikidata.org/wiki/Q199804 |
| concepts[3].display_name | COPD |
| concepts[4].id | https://openalex.org/C168563851 |
| concepts[4].level | 2 |
| concepts[4].score | 0.5642386674880981 |
| concepts[4].wikidata | https://www.wikidata.org/wiki/Q1436668 |
| concepts[4].display_name | Randomized controlled trial |
| concepts[5].id | https://openalex.org/C535046627 |
| concepts[5].level | 2 |
| concepts[5].score | 0.4921781122684479 |
| concepts[5].wikidata | https://www.wikidata.org/wiki/Q30612 |
| concepts[5].display_name | Clinical trial |
| concepts[6].id | https://openalex.org/C2780861865 |
| concepts[6].level | 3 |
| concepts[6].score | 0.448725163936615 |
| concepts[6].wikidata | https://www.wikidata.org/wiki/Q4677923 |
| concepts[6].display_name | Acute exacerbation of chronic obstructive pulmonary disease |
| concepts[7].id | https://openalex.org/C177713679 |
| concepts[7].level | 1 |
| concepts[7].score | 0.37604162096977234 |
| concepts[7].wikidata | https://www.wikidata.org/wiki/Q679690 |
| concepts[7].display_name | Intensive care medicine |
| concepts[8].id | https://openalex.org/C1862650 |
| concepts[8].level | 1 |
| concepts[8].score | 0.37553492188453674 |
| concepts[8].wikidata | https://www.wikidata.org/wiki/Q186005 |
| concepts[8].display_name | Physical therapy |
| concepts[9].id | https://openalex.org/C126322002 |
| concepts[9].level | 1 |
| concepts[9].score | 0.3581346869468689 |
| concepts[9].wikidata | https://www.wikidata.org/wiki/Q11180 |
| concepts[9].display_name | Internal medicine |
| concepts[10].id | https://openalex.org/C204787440 |
| concepts[10].level | 2 |
| concepts[10].score | 0.203322172164917 |
| concepts[10].wikidata | https://www.wikidata.org/wiki/Q188504 |
| concepts[10].display_name | Alternative medicine |
| concepts[11].id | https://openalex.org/C142724271 |
| concepts[11].level | 1 |
| concepts[11].score | 0.0 |
| concepts[11].wikidata | https://www.wikidata.org/wiki/Q7208 |
| concepts[11].display_name | Pathology |
| keywords[0].id | https://openalex.org/keywords/medicine |
| keywords[0].score | 0.9346504211425781 |
| keywords[0].display_name | Medicine |
| keywords[1].id | https://openalex.org/keywords/exacerbation |
| keywords[1].score | 0.7290292978286743 |
| keywords[1].display_name | Exacerbation |
| keywords[2].id | https://openalex.org/keywords/placebo |
| keywords[2].score | 0.7018404006958008 |
| keywords[2].display_name | Placebo |
| keywords[3].id | https://openalex.org/keywords/copd |
| keywords[3].score | 0.6238923668861389 |
| keywords[3].display_name | COPD |
| keywords[4].id | https://openalex.org/keywords/randomized-controlled-trial |
| keywords[4].score | 0.5642386674880981 |
| keywords[4].display_name | Randomized controlled trial |
| keywords[5].id | https://openalex.org/keywords/clinical-trial |
| keywords[5].score | 0.4921781122684479 |
| keywords[5].display_name | Clinical trial |
| keywords[6].id | https://openalex.org/keywords/acute-exacerbation-of-chronic-obstructive-pulmonary-disease |
| keywords[6].score | 0.448725163936615 |
| keywords[6].display_name | Acute exacerbation of chronic obstructive pulmonary disease |
| keywords[7].id | https://openalex.org/keywords/intensive-care-medicine |
| keywords[7].score | 0.37604162096977234 |
| keywords[7].display_name | Intensive care medicine |
| keywords[8].id | https://openalex.org/keywords/physical-therapy |
| keywords[8].score | 0.37553492188453674 |
| keywords[8].display_name | Physical therapy |
| keywords[9].id | https://openalex.org/keywords/internal-medicine |
| keywords[9].score | 0.3581346869468689 |
| keywords[9].display_name | Internal medicine |
| keywords[10].id | https://openalex.org/keywords/alternative-medicine |
| keywords[10].score | 0.203322172164917 |
| keywords[10].display_name | Alternative medicine |
| language | en |
| locations[0].id | doi:10.21203/rs.3.rs-1918642/v1 |
| locations[0].is_oa | True |
| locations[0].source.id | https://openalex.org/S4306402450 |
| locations[0].source.issn | |
| locations[0].source.type | repository |
| locations[0].source.is_oa | False |
| locations[0].source.issn_l | |
| locations[0].source.is_core | False |
| locations[0].source.is_in_doaj | False |
| locations[0].source.display_name | Research Square (Research Square) |
| locations[0].source.host_organization | https://openalex.org/I4210096694 |
| locations[0].source.host_organization_name | Research Square (United States) |
| locations[0].source.host_organization_lineage | https://openalex.org/I4210096694 |
| locations[0].license | cc-by |
| locations[0].pdf_url | https://www.researchsquare.com/article/rs-1918642/latest.pdf |
| locations[0].version | acceptedVersion |
| locations[0].raw_type | posted-content |
| locations[0].license_id | https://openalex.org/licenses/cc-by |
| locations[0].is_accepted | True |
| locations[0].is_published | False |
| locations[0].raw_source_name | |
| locations[0].landing_page_url | https://doi.org/10.21203/rs.3.rs-1918642/v1 |
| indexed_in | crossref |
| authorships[0].author.id | https://openalex.org/A5034924680 |
| authorships[0].author.orcid | https://orcid.org/0000-0002-3143-7999 |
| authorships[0].author.display_name | Xiao-Yang Hu |
| authorships[0].countries | GB |
| authorships[0].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[0].affiliations[0].raw_affiliation_string | University of Southampton Faculty of Medicine |
| authorships[0].institutions[0].id | https://openalex.org/I43439940 |
| authorships[0].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[0].institutions[0].type | education |
| authorships[0].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[0].institutions[0].country_code | GB |
| authorships[0].institutions[0].display_name | University of Southampton |
| authorships[0].author_position | first |
| authorships[0].raw_author_name | Xiao-Yang Hu |
| authorships[0].is_corresponding | False |
| authorships[0].raw_affiliation_strings | University of Southampton Faculty of Medicine |
| authorships[1].author.id | https://openalex.org/A5005648976 |
| authorships[1].author.orcid | |
| authorships[1].author.display_name | Tom Oliver |
| authorships[1].countries | GB |
| authorships[1].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[1].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[1].institutions[0].id | https://openalex.org/I43439940 |
| authorships[1].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[1].institutions[0].type | education |
| authorships[1].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[1].institutions[0].country_code | GB |
| authorships[1].institutions[0].display_name | University of Southampton |
| authorships[1].author_position | middle |
| authorships[1].raw_author_name | Tom Oliver |
| authorships[1].is_corresponding | False |
| authorships[1].raw_affiliation_strings | University of Southampton |
| authorships[2].author.id | https://openalex.org/A5019357161 |
| authorships[2].author.orcid | https://orcid.org/0000-0002-5227-3444 |
| authorships[2].author.display_name | Merlin Willcox |
| authorships[2].countries | GB |
| authorships[2].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[2].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[2].institutions[0].id | https://openalex.org/I43439940 |
| authorships[2].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[2].institutions[0].type | education |
| authorships[2].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[2].institutions[0].country_code | GB |
| authorships[2].institutions[0].display_name | University of Southampton |
| authorships[2].author_position | middle |
| authorships[2].raw_author_name | Merlin Willcox |
| authorships[2].is_corresponding | False |
| authorships[2].raw_affiliation_strings | University of Southampton |
| authorships[3].author.id | https://openalex.org/A5110174311 |
| authorships[3].author.orcid | |
| authorships[3].author.display_name | Catherine Simpson |
| authorships[3].countries | GB |
| authorships[3].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[3].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[3].institutions[0].id | https://openalex.org/I43439940 |
| authorships[3].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[3].institutions[0].type | education |
| authorships[3].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[3].institutions[0].country_code | GB |
| authorships[3].institutions[0].display_name | University of Southampton |
| authorships[3].author_position | middle |
| authorships[3].raw_author_name | Catherine Simpson |
| authorships[3].is_corresponding | False |
| authorships[3].raw_affiliation_strings | University of Southampton |
| authorships[4].author.id | https://openalex.org/A5035379938 |
| authorships[4].author.orcid | https://orcid.org/0000-0003-1905-5610 |
| authorships[4].author.display_name | Kerensa Thorne |
| authorships[4].countries | GB |
| authorships[4].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[4].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[4].institutions[0].id | https://openalex.org/I43439940 |
| authorships[4].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[4].institutions[0].type | education |
| authorships[4].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[4].institutions[0].country_code | GB |
| authorships[4].institutions[0].display_name | University of Southampton |
| authorships[4].author_position | middle |
| authorships[4].raw_author_name | Kerensa Thorne |
| authorships[4].is_corresponding | False |
| authorships[4].raw_affiliation_strings | University of Southampton |
| authorships[5].author.id | https://openalex.org/A5010076340 |
| authorships[5].author.orcid | https://orcid.org/0000-0003-1075-4378 |
| authorships[5].author.display_name | Jeanne Trill |
| authorships[5].countries | GB |
| authorships[5].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[5].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[5].institutions[0].id | https://openalex.org/I43439940 |
| authorships[5].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[5].institutions[0].type | education |
| authorships[5].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[5].institutions[0].country_code | GB |
| authorships[5].institutions[0].display_name | University of Southampton |
| authorships[5].author_position | middle |
| authorships[5].raw_author_name | Jeanne Trill |
| authorships[5].is_corresponding | False |
| authorships[5].raw_affiliation_strings | University of Southampton |
| authorships[6].author.id | https://openalex.org/A5011449104 |
| authorships[6].author.orcid | https://orcid.org/0000-0001-8939-7312 |
| authorships[6].author.display_name | Nick Francis |
| authorships[6].countries | GB |
| authorships[6].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[6].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[6].institutions[0].id | https://openalex.org/I43439940 |
| authorships[6].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[6].institutions[0].type | education |
| authorships[6].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[6].institutions[0].country_code | GB |
| authorships[6].institutions[0].display_name | University of Southampton |
| authorships[6].author_position | middle |
| authorships[6].raw_author_name | Nick Francis |
| authorships[6].is_corresponding | False |
| authorships[6].raw_affiliation_strings | University of Southampton |
| authorships[7].author.id | https://openalex.org/A5091500770 |
| authorships[7].author.orcid | https://orcid.org/0000-0001-5432-7437 |
| authorships[7].author.display_name | Beth Stuart |
| authorships[7].countries | GB |
| authorships[7].affiliations[0].institution_ids | https://openalex.org/I166337079 |
| authorships[7].affiliations[0].raw_affiliation_string | Queen Mary University of London |
| authorships[7].institutions[0].id | https://openalex.org/I166337079 |
| authorships[7].institutions[0].ror | https://ror.org/026zzn846 |
| authorships[7].institutions[0].type | education |
| authorships[7].institutions[0].lineage | https://openalex.org/I124357947, https://openalex.org/I166337079 |
| authorships[7].institutions[0].country_code | GB |
| authorships[7].institutions[0].display_name | Queen Mary University of London |
| authorships[7].author_position | middle |
| authorships[7].raw_author_name | Beth Stuart |
| authorships[7].is_corresponding | False |
| authorships[7].raw_affiliation_strings | Queen Mary University of London |
| authorships[8].author.id | https://openalex.org/A5077591623 |
| authorships[8].author.orcid | https://orcid.org/0000-0001-5939-1155 |
| authorships[8].author.display_name | Mike Thomas |
| authorships[8].countries | GB |
| authorships[8].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[8].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[8].institutions[0].id | https://openalex.org/I43439940 |
| authorships[8].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[8].institutions[0].type | education |
| authorships[8].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[8].institutions[0].country_code | GB |
| authorships[8].institutions[0].display_name | University of Southampton |
| authorships[8].author_position | middle |
| authorships[8].raw_author_name | Michael Thomas |
| authorships[8].is_corresponding | False |
| authorships[8].raw_affiliation_strings | University of Southampton |
| authorships[9].author.id | https://openalex.org/A5029134034 |
| authorships[9].author.orcid | https://orcid.org/0000-0003-3664-1873 |
| authorships[9].author.display_name | Paul Little |
| authorships[9].countries | GB |
| authorships[9].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[9].affiliations[0].raw_affiliation_string | University of Southampton Faculty of Medicine |
| authorships[9].institutions[0].id | https://openalex.org/I43439940 |
| authorships[9].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[9].institutions[0].type | education |
| authorships[9].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[9].institutions[0].country_code | GB |
| authorships[9].institutions[0].display_name | University of Southampton |
| authorships[9].author_position | middle |
| authorships[9].raw_author_name | Paul Little |
| authorships[9].is_corresponding | False |
| authorships[9].raw_affiliation_strings | University of Southampton Faculty of Medicine |
| authorships[10].author.id | https://openalex.org/A5100317669 |
| authorships[10].author.orcid | https://orcid.org/0000-0002-0320-061X |
| authorships[10].author.display_name | Jianping Liu |
| authorships[10].countries | CN |
| authorships[10].affiliations[0].institution_ids | https://openalex.org/I17747738 |
| authorships[10].affiliations[0].raw_affiliation_string | Beijing University of Chinese Medicine |
| authorships[10].institutions[0].id | https://openalex.org/I17747738 |
| authorships[10].institutions[0].ror | https://ror.org/05damtm70 |
| authorships[10].institutions[0].type | education |
| authorships[10].institutions[0].lineage | https://openalex.org/I17747738 |
| authorships[10].institutions[0].country_code | CN |
| authorships[10].institutions[0].display_name | Beijing University of Chinese Medicine |
| authorships[10].author_position | middle |
| authorships[10].raw_author_name | Jian-Ping Liu |
| authorships[10].is_corresponding | False |
| authorships[10].raw_affiliation_strings | Beijing University of Chinese Medicine |
| authorships[11].author.id | https://openalex.org/A5084659683 |
| authorships[11].author.orcid | https://orcid.org/0000-0002-9579-8021 |
| authorships[11].author.display_name | Gareth Griffiths |
| authorships[11].countries | GB |
| authorships[11].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[11].affiliations[0].raw_affiliation_string | University of Southampton |
| authorships[11].institutions[0].id | https://openalex.org/I43439940 |
| authorships[11].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[11].institutions[0].type | education |
| authorships[11].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[11].institutions[0].country_code | GB |
| authorships[11].institutions[0].display_name | University of Southampton |
| authorships[11].author_position | middle |
| authorships[11].raw_author_name | Gareth Griffiths |
| authorships[11].is_corresponding | False |
| authorships[11].raw_affiliation_strings | University of Southampton |
| authorships[12].author.id | https://openalex.org/A5053346837 |
| authorships[12].author.orcid | https://orcid.org/0000-0002-5127-4509 |
| authorships[12].author.display_name | Michael Moore |
| authorships[12].countries | GB |
| authorships[12].affiliations[0].institution_ids | https://openalex.org/I43439940 |
| authorships[12].affiliations[0].raw_affiliation_string | University of Southampton Faculty of Medicine |
| authorships[12].institutions[0].id | https://openalex.org/I43439940 |
| authorships[12].institutions[0].ror | https://ror.org/01ryk1543 |
| authorships[12].institutions[0].type | education |
| authorships[12].institutions[0].lineage | https://openalex.org/I43439940 |
| authorships[12].institutions[0].country_code | GB |
| authorships[12].institutions[0].display_name | University of Southampton |
| authorships[12].author_position | last |
| authorships[12].raw_author_name | Michael Moore |
| authorships[12].is_corresponding | False |
| authorships[12].raw_affiliation_strings | University of Southampton Faculty of Medicine |
| has_content.pdf | True |
| has_content.grobid_xml | True |
| is_paratext | False |
| open_access.is_oa | True |
| open_access.oa_url | https://www.researchsquare.com/article/rs-1918642/latest.pdf |
| open_access.oa_status | green |
| open_access.any_repository_has_fulltext | False |
| created_date | 2025-10-10T00:00:00 |
| display_name | Treating Acute EXacerbation of COPD with Chinese HerbAL MedIcine to aid AntiBiotic Use Reduction (EXCALIBUR): study protocol of a randomised double-blind, placebo-controlled feasibility trial |
| has_fulltext | False |
| is_retracted | False |
| updated_date | 2025-11-06T03:46:38.306776 |
| primary_topic.id | https://openalex.org/T10143 |
| primary_topic.field.id | https://openalex.org/fields/27 |
| primary_topic.field.display_name | Medicine |
| primary_topic.score | 0.9979000091552734 |
| primary_topic.domain.id | https://openalex.org/domains/4 |
| primary_topic.domain.display_name | Health Sciences |
| primary_topic.subfield.id | https://openalex.org/subfields/2740 |
| primary_topic.subfield.display_name | Pulmonary and Respiratory Medicine |
| primary_topic.display_name | Chronic Obstructive Pulmonary Disease (COPD) Research |
| related_works | https://openalex.org/W3031564007, https://openalex.org/W2069237071, https://openalex.org/W2409754429, https://openalex.org/W3184513407, https://openalex.org/W2762415100, https://openalex.org/W2312821885, https://openalex.org/W2077638660, https://openalex.org/W2160736681, https://openalex.org/W2618090565, https://openalex.org/W2313936276 |
| cited_by_count | 0 |
| locations_count | 1 |
| best_oa_location.id | doi:10.21203/rs.3.rs-1918642/v1 |
| best_oa_location.is_oa | True |
| best_oa_location.source.id | https://openalex.org/S4306402450 |
| best_oa_location.source.issn | |
| best_oa_location.source.type | repository |
| best_oa_location.source.is_oa | False |
| best_oa_location.source.issn_l | |
| best_oa_location.source.is_core | False |
| best_oa_location.source.is_in_doaj | False |
| best_oa_location.source.display_name | Research Square (Research Square) |
| best_oa_location.source.host_organization | https://openalex.org/I4210096694 |
| best_oa_location.source.host_organization_name | Research Square (United States) |
| best_oa_location.source.host_organization_lineage | https://openalex.org/I4210096694 |
| best_oa_location.license | cc-by |
| best_oa_location.pdf_url | https://www.researchsquare.com/article/rs-1918642/latest.pdf |
| best_oa_location.version | acceptedVersion |
| best_oa_location.raw_type | posted-content |
| best_oa_location.license_id | https://openalex.org/licenses/cc-by |
| best_oa_location.is_accepted | True |
| best_oa_location.is_published | False |
| best_oa_location.raw_source_name | |
| best_oa_location.landing_page_url | https://doi.org/10.21203/rs.3.rs-1918642/v1 |
| primary_location.id | doi:10.21203/rs.3.rs-1918642/v1 |
| primary_location.is_oa | True |
| primary_location.source.id | https://openalex.org/S4306402450 |
| primary_location.source.issn | |
| primary_location.source.type | repository |
| primary_location.source.is_oa | False |
| primary_location.source.issn_l | |
| primary_location.source.is_core | False |
| primary_location.source.is_in_doaj | False |
| primary_location.source.display_name | Research Square (Research Square) |
| primary_location.source.host_organization | https://openalex.org/I4210096694 |
| primary_location.source.host_organization_name | Research Square (United States) |
| primary_location.source.host_organization_lineage | https://openalex.org/I4210096694 |
| primary_location.license | cc-by |
| primary_location.pdf_url | https://www.researchsquare.com/article/rs-1918642/latest.pdf |
| primary_location.version | acceptedVersion |
| primary_location.raw_type | posted-content |
| primary_location.license_id | https://openalex.org/licenses/cc-by |
| primary_location.is_accepted | True |
| primary_location.is_published | False |
| primary_location.raw_source_name | |
| primary_location.landing_page_url | https://doi.org/10.21203/rs.3.rs-1918642/v1 |
| publication_date | 2022-08-30 |
| publication_year | 2022 |
| referenced_works | https://openalex.org/W3033438356, https://openalex.org/W2797313070, https://openalex.org/W2168630917, https://openalex.org/W2020356839, https://openalex.org/W2957912127, https://openalex.org/W2122147693, https://openalex.org/W1889806553, https://openalex.org/W2038909459, https://openalex.org/W2742836413, https://openalex.org/W2009596210, https://openalex.org/W3147005240, https://openalex.org/W4283798163, https://openalex.org/W4213438448, https://openalex.org/W4210315185, https://openalex.org/W2123689455, https://openalex.org/W3027006304, https://openalex.org/W4200537010, https://openalex.org/W2166023752, https://openalex.org/W2738590667, https://openalex.org/W2138664283 |
| referenced_works_count | 20 |
| abstract_inverted_index.1 | 262 |
| abstract_inverted_index.: | 254 |
| abstract_inverted_index.a | 11, 67, 100, 115, 144, 237 |
| abstract_inverted_index.12 | 202 |
| abstract_inverted_index.14 | 194 |
| abstract_inverted_index.GP | 159 |
| abstract_inverted_index.If | 221 |
| abstract_inverted_index.UK | 33 |
| abstract_inverted_index.We | 170 |
| abstract_inverted_index.be | 166 |
| abstract_inverted_index.by | 57, 131 |
| abstract_inverted_index.in | 16, 22, 32, 38, 109, 249 |
| abstract_inverted_index.is | 43, 66, 114, 160, 257 |
| abstract_inverted_index.of | 4, 27, 40, 45, 52, 89, 99, 163, 210, 217, 227, 242, 247 |
| abstract_inverted_index.on | 107, 261 |
| abstract_inverted_index.or | 153, 191 |
| abstract_inverted_index.st | 263 |
| abstract_inverted_index.to | 69, 83, 95, 168, 172, 181, 198 |
| abstract_inverted_index.up | 200 |
| abstract_inverted_index.we | 232 |
| abstract_inverted_index.70% | 26 |
| abstract_inverted_index.The | 73, 204 |
| abstract_inverted_index.aim | 171 |
| abstract_inverted_index.and | 24, 87, 155, 197, 214, 230 |
| abstract_inverted_index.are | 10, 36, 54 |
| abstract_inverted_index.for | 14, 63, 156, 193, 201, 236, 244 |
| abstract_inverted_index.has | 80 |
| abstract_inverted_index.not | 55 |
| abstract_inverted_index.per | 177 |
| abstract_inverted_index.the | 46, 50, 81, 97, 132, 158, 184, 208, 215, 225, 245 |
| abstract_inverted_index.use | 162 |
| abstract_inverted_index.via | 259 |
| abstract_inverted_index.COPD | 64 |
| abstract_inverted_index.III, | 117 |
| abstract_inverted_index.July | 264 |
| abstract_inverted_index.SFJD | 108, 189, 243 |
| abstract_inverted_index.This | 92, 112, 255 |
| abstract_inverted_index.aged | 139 |
| abstract_inverted_index.aims | 94 |
| abstract_inverted_index.care | 35 |
| abstract_inverted_index.days | 196 |
| abstract_inverted_index.old, | 142 |
| abstract_inverted_index.part | 44 |
| abstract_inverted_index.seek | 234 |
| abstract_inverted_index.test | 96 |
| abstract_inverted_index.them | 180 |
| abstract_inverted_index.this | 222 |
| abstract_inverted_index.unit | 136 |
| abstract_inverted_index.use. | 72 |
| abstract_inverted_index.whom | 157 |
| abstract_inverted_index.will | 165, 233 |
| abstract_inverted_index.with | 30, 126, 143, 148 |
| abstract_inverted_index.2021, | 265 |
| abstract_inverted_index.Acute | 2 |
| abstract_inverted_index.Trial | 252 |
| abstract_inverted_index.care, | 18 |
| abstract_inverted_index.care. | 48, 251 |
| abstract_inverted_index.fully | 238 |
| abstract_inverted_index.major | 12 |
| abstract_inverted_index.phase | 116 |
| abstract_inverted_index.seven | 174 |
| abstract_inverted_index.stay. | 91 |
| abstract_inverted_index.study | 93, 113, 212 |
| abstract_inverted_index.trial | 106, 125, 135, 218, 223, 241, 256 |
| abstract_inverted_index.which | 42 |
| abstract_inverted_index.years | 141 |
| abstract_inverted_index.≥40 | 140 |
| abstract_inverted_index.(SFJD) | 79 |
| abstract_inverted_index.AECOPD | 31, 248 |
| abstract_inverted_index.ISRCTN | 260 |
| abstract_inverted_index.blind, | 103, 121 |
| abstract_inverted_index.caused | 56 |
| abstract_inverted_index.double | 102, 120 |
| abstract_inverted_index.either | 183 |
| abstract_inverted_index.follow | 199 |
| abstract_inverted_index.herbal | 75, 187 |
| abstract_inverted_index.month, | 178 |
| abstract_inverted_index.nested | 127 |
| abstract_inverted_index.patent | 185 |
| abstract_inverted_index.reason | 13 |
| abstract_inverted_index.reduce | 70, 84 |
| abstract_inverted_index.sputum | 150 |
| abstract_inverted_index.study, | 129 |
| abstract_inverted_index.weeks. | 203 |
| abstract_inverted_index.(SCTU). | 137 |
| abstract_inverted_index.AECOPD, | 146 |
| abstract_inverted_index.AECOPD. | 110 |
| abstract_inverted_index.Chinese | 74, 186 |
| abstract_inverted_index.Finding | 59 |
| abstract_inverted_index.Jieduâ | 78 |
| abstract_inverted_index.Shufeng | 77 |
| abstract_inverted_index.chronic | 5 |
| abstract_inverted_index.current | 145 |
| abstract_inverted_index.disease | 8 |
| abstract_inverted_index.failure | 86 |
| abstract_inverted_index.funding | 235 |
| abstract_inverted_index.include | 207 |
| abstract_inverted_index.placebo | 192 |
| abstract_inverted_index.powered | 239 |
| abstract_inverted_index.primary | 17, 34, 205, 250 |
| abstract_inverted_index.receipt | 39 |
| abstract_inverted_index.receive | 182 |
| abstract_inverted_index.recruit | 173 |
| abstract_inverted_index.two-arm | 118 |
| abstract_inverted_index.volume, | 152 |
| abstract_inverted_index.(AECOPD) | 9 |
| abstract_inverted_index.Abstract | 0 |
| abstract_inverted_index.However, | 49 |
| abstract_inverted_index.Methods: | 111 |
| abstract_inverted_index.Patients | 138 |
| abstract_inverted_index.capsules | 190 |
| abstract_inverted_index.clinical | 105, 134 |
| abstract_inverted_index.delivery | 229 |
| abstract_inverted_index.diaries. | 219 |
| abstract_inverted_index.duration | 88 |
| abstract_inverted_index.eligible | 167, 175 |
| abstract_inverted_index.hospital | 19, 90 |
| abstract_inverted_index.majority | 51 |
| abstract_inverted_index.medicine | 76, 188 |
| abstract_inverted_index.outcomes | 206 |
| abstract_inverted_index.patients | 28 |
| abstract_inverted_index.priority | 68 |
| abstract_inverted_index.standard | 47 |
| abstract_inverted_index.bacteria. | 58 |
| abstract_inverted_index.currently | 37 |
| abstract_inverted_index.effective | 60 |
| abstract_inverted_index.function, | 23 |
| abstract_inverted_index.increased | 149 |
| abstract_inverted_index.potential | 82 |
| abstract_inverted_index.pulmonary | 7 |
| abstract_inverted_index.randomise | 179 |
| abstract_inverted_index.treatment | 85, 246 |
| abstract_inverted_index.antibiotic | 71 |
| abstract_inverted_index.completion | 216 |
| abstract_inverted_index.follow-up, | 231 |
| abstract_inverted_index.mortality. | 25 |
| abstract_inverted_index.presenting | 29, 147 |
| abstract_inverted_index.purulence/ | 151 |
| abstract_inverted_index.registered | 258 |
| abstract_inverted_index.retention, | 213 |
| abstract_inverted_index.treatments | 62 |
| abstract_inverted_index.Background: | 1 |
| abstract_inverted_index.Discussion: | 220 |
| abstract_inverted_index.Southampton | 133 |
| abstract_inverted_index.admissions, | 20 |
| abstract_inverted_index.antibiotics | 41 |
| abstract_inverted_index.consecutive | 195 |
| abstract_inverted_index.considering | 161 |
| abstract_inverted_index.coordinated | 130 |
| abstract_inverted_index.feasibility | 98, 124, 209, 226 |
| abstract_inverted_index.identifier: | 266 |
| abstract_inverted_index.obstructive | 6 |
| abstract_inverted_index.qualitative | 128 |
| abstract_inverted_index.randomised, | 101, 122 |
| abstract_inverted_index.antibiotics, | 164 |
| abstract_inverted_index.demonstrates | 224 |
| abstract_inverted_index.exacerbation | 3 |
| abstract_inverted_index.individually | 119 |
| abstract_inverted_index.participants | 176 |
| abstract_inverted_index.participate. | 169 |
| abstract_inverted_index.recruitment, | 211, 228 |
| abstract_inverted_index.registration | 253 |
| abstract_inverted_index.consultations | 15 |
| abstract_inverted_index.deterioration | 21 |
| abstract_inverted_index.exacerbations | 53, 65 |
| abstract_inverted_index.non-antibiotic | 61 |
| abstract_inverted_index.ISRCTN26614726. | 267 |
| abstract_inverted_index.breathlessness, | 154 |
| abstract_inverted_index.placebo-controlled | 104, 123, 240 |
| cited_by_percentile_year | |
| countries_distinct_count | 2 |
| institutions_distinct_count | 13 |
| sustainable_development_goals[0].id | https://metadata.un.org/sdg/3 |
| sustainable_development_goals[0].score | 0.800000011920929 |
| sustainable_development_goals[0].display_name | Good health and well-being |
| citation_normalized_percentile.value | 0.19291346 |
| citation_normalized_percentile.is_in_top_1_percent | False |
| citation_normalized_percentile.is_in_top_10_percent | False |