 Supercapacitor
Swipe Supercapacitor Vs...
Supercapacitor
Swipe Supercapacitor Vs...

Supercapacitor News
Description
A supercapacitor ( SC ), also called an ultracapacitor , is a high-capacity capacitor, with a capacitance value much higher than solid-state capacitors but with lower voltage limits. It bridges the gap between electrolytic capacitors and rechargeable batteries. It typically stores 10 to 100 times more energy per unit volume or mass than electrolytic capacitors, can accept and deliver charge much faster than batteries, and tolerates many more charge and discharge cycles than rechargeable batteries.
Supercapacitors are used in applications requiring many rapid charge/discharge cycles, rather than long-term compact energy storage — in automobiles, buses, trains, cranes and elevators, where they are used for regenerative braking, short-term energy storage, or burst-mode power delivery. Smaller units are used as power backup for static random-access memory (SRAM).
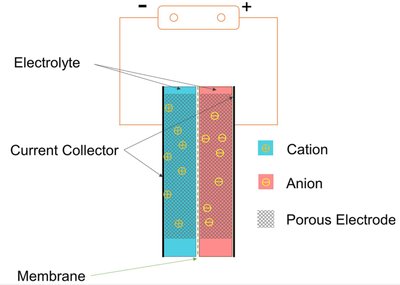
Unlike ordinary capacitors, supercapacitors do not use the conventional solid dielectric, but rather, they use double-layer capacitance on one electrode and electrochemical battery electrode as the other. The electrochemical charge storage mechanisms in solid media can be roughly (there is an overlap in some systems) classified into 3 types:
- Electrostatic double-layer capacitors ( EDLCs ) use carbon electrodes or derivatives with much higher electrostatic double-layer capacitance than electrochemical pseudocapacitance, achieving separation of charge in a Helmholtz double layer at the interface between the surface of a conductive electrode and an electrolyte. The separation of charge is of the order of a few ångströms (0.3–0.8 nm), much smaller than in a conventional capacitor. The electric charge is EDLCs is stored in a two-dimensional interphase (surface) of an electronic conductor (e.g. carbon particle) and ionic conductor (electrolyte solution).
- Batteries with solid electroactive materials store charge in bulk solid phases by virtue of redox chemical reactions.
- Electrochemical supercapacitors (ECSCs) fall in between EDLs and batteries. ECSCs use metal oxide or conducting polymer electrodes with a high amount of electrochemical pseudocapacitance additional to the double-layer capacitance. Pseudocapacitance is achieved by Faradaic electron charge-transfer with redox reactions, intercalation or electrosorption.
In solid-state capacitors, the mobile charges are electrons, and the gap between electrodes is a layer of a dielectric. In electrochemical double-layer capacitors, the mobile charges are solvated ions (cations and anions), and the effective thickness is determined on each of the two electrodes by their electrochemical double layer structure. In batteries the charge is stored in the bulk volume of solid phases, which have both electronic and ionic conductivities. In electrochemical supercapacitors, the charge storage mechanisms either combine the double-layer and battery mechanisms, or are based on mechanisms, which are intermediate between true double layer and true battery.
Related
MoreCollections
No collections available for this topic.
Details
- Slug: supercapacitor
- Total Views: 753
- Added: Jul 20, 2024